GLUCAN
A Bibliographic Collection
GLUCAN
Immunostimulant Activity
Glucans are natural polysaccharides compounded by glucopyranosic units and can be
found in grain, seaweed, cell wall of bacteria and cell wall of fungi.
Glucans derived from various types of grain, are essential components of human diet but
it was determined that purified Beta-Glucans, specifically Beta 1-3 Glucan, solubilized
from yeast, are strong stimulants of the immune system1.
They have the capacity to activate macrophages, neutrophils, and other cells that
possess specific receptors for these compounds. The activation of these cells by glucan
stimulates the body’s non-specific immune response.
Other polysaccharides, like as mannans and Alfa 1-4 or Beta 1-4 polymers of glucose
don’t possess this activity. Glucan strengthens the body’s defence mechanism under
various conditions. It was demonstrated that the reason for this strengthening is due to
the increased rate of phagocytosis through glucan-activated macrophages2.
1
Kokashis, P.L., Williams, D.L., Cook, J.A. and Di Luzio, N.R.
Increased resistance to Staphylococcus aureus infection and enhancement in serum lysozyme activity by glucan.
Science 199, 1340-1342 (1978).
2
Sherwood, E.R., Williams, D.l. and N.R. Di Luzio.
Glucan stimulates production of antitumor cytolytic cytostatic factors by macrophages. J. Biol. Resp. Mod- 6, 358381 (1986)
2
Management Of Adenoid Hypertrophy: Efficacy Of
Beta -Glucan
La terapia dell’ipertrofia delle adenoidi: efficacia del
beta-glucano
The medical treatmem of the adenoid hypertrophy
cannot put aside from an immunostimulant
theraphy. We report the results of a placebo
"controlled, randomized,double-blind study on the
efficacy and the safety of beta-glucan. A total of
103 pediatric patients (middle age 6,5 years) were
enrolled in our study between September 2000 and
December 2001. The selected patients were
affected from odenoid hypertrophy of 3°(68%) and
4° (32%) degree. The elegibility criteria including:
1) a documented diagnosis of adenoid hypertrophy;
2} not allergy; 3} any benefit from precedent
therapies; 4) common environment conditions of
life. The patients treated with beta-glucan (group
A) had a reduction of adenoid hypertrophy of
62,5% with improvement of signs and symptoms,
whereas the patients that received placebo (group
B) had an improvement of adenoid hypertrophy of
13,1% (p<0,05). Our study shows that the
administration of beta-glucan reduces the adenoid
hypertrophy and improves nasal functions. The
beta-glucan, with the immunostimulant action,
that is expounded with the increase of lymphocyte
B and T and with the incentive to macrophages
activity,
improves
the
trophism
and
the
immunologic action of Waldeyer's ring, particularly
of the vegetations adenoids.
Il trattamento medico dell’ipertrofia delle adenoidi non può
prescindere da una terapia immunostimolante. Riportiamo i
risultati di uno studio controllato con placebo, randomizzato
e in doppio cieco riguardo l’efficacia e la sicurezza del betaglucano. Per il nostro studio è stato arruolato un totale di
103 pazienti pediatrici (età media 6.5 anni), tra il settembre
2000 ed il dicembre 2001. I pazienti selezionati erano affetti
da ipertrofia delle adenoidi di 3° (68 %) e di 4° grado (32
%). I criteri di selezione hanno considerato: 1) una diagnosi
documentata di ipertrofia delle adenoidi; 2) l’assenza di
allergie; 3) assenza di benefici da precedenti terapie; 4)
condizioni ambientali di vita comuni. I pazienti trattati col
beta-glucano (gruppo A) hanno presentato una riduzione
dell’ipertrofia delle adenoidi del 62.5 % con miglioramento
dei segni e dei sintomi, mentre i pazienti che hanno ricevuto
il placebo (gruppo B) hanno avuto un miglioramento
dell’ipertrofia delle adenoidi del 13.1 % (p<0.05). Il nostro
studio mostra che la somministrazione del beta-glucano
riduce l’ipertrofia delle adenoidi e migliora la funzionalità
nasale. Il beta-glucano, con la sua azione immunostimolante,
che si manifesta con l’aumento di linfociti B e T e con
l’incremento dell’attività macrofagica, migliora il trofismo e
l’azione
immunologica
dell’anello
di
Waldeyer,
particolarmente della vegetazione adenoide.
Tricarico, D; Ascione, E; Avvisati, F; Caterino, R;Varricchio,
A; Imperiali, M, Italy
Tricarico D; Ascione E; Avvisati F; Caterino R; Varricchio A;
Imperiali M
Immunostimulant oxidized beta-glucan conjugates.
Beta-glucani coniugati ossidati immunostimolanti.
Cross GG; Jennings HJ; Whitfield DM; Penney CL; Zacharie B; Gagnon
L Int Immunopharmacol 2001 Mar;1(3):539-50 (ISSN: 1567-5769)
National Research Council, 100 Sussex Drive, Ottawa, Ontario, Canada
K1A 0R6.
Cross GG; Jennings HJ; Whitfield DM; Penney CL; Zacharie B;
Gagnon L Int Immunopharmacol 2001 Mar;1(3):539-50 (ISSN:
1567-5769) National Research Council, 100 Sussex Drive, Ottawa,
Ontario, Canada K1A 0R6.
I beta-glucani sono polisaccaridi che agiscono come stimolanti non
specifici del sistema immunitario. Molti beta-glucani sono
moderatamente solubili in acqua. Questo lavoro descrive una
procedura ossidativa che solubilizza il beta-glucano ricavato da
Saccaromyces
cerevisiae
mantenendo
le
sue
proprietà
immunostimolatorie. Inoltre, i carbossilati presenti ai siti di
ossidazione consentono la coniugazione di immunostimolanti a
molecola piccola. Sia il beta-glucano ossidato precursore che i suoi
coniugati
con
O-beta-alanil-5-[6-(N,N’-dietilamino)purin-9yl]pentanolo, stimolano i linfociti T citotossici (CTL), le cellule B e i
macrofagi. Inoltre entrambi stimolano le cellule “natural killer” (NK),
proprietà che la purina a molecola piccola non possiede.
Beta-Glucans are polysaccharides that act as nonspecific immune
system stimulants. However, many beta-Glucans are sparingly
soluble in water. This work describes an oxidative procedure,
which solubilizes the beta-Glucan from Saccharomyces cerevisiae
and maintains its immunostimulatory properties. Furthermore, the
carboxylates at the site of oxidation allow for the conjugation of
small molecule immunostimulants. Both the parent oxidized betaglucan and its conjugates with O-beta-alanyl-5-[6-(N,N'dimethylamino)purin-9-yl]pentanol
stimulate
cytotoxic
Tlymphocytes (CTLs), B cells and macrophages. In addition, they
both stimulate natural killer (NK) cells, a property which the small
molecule purine does not possess.
3
Effect of glucan on granulopoiesis and macrophage
genesis in mice
Effetto del glucano sulla granulopoiesi
generazione di macrofagi nel topo
C Burgaleta and DW Golde Cancer Research; 37: 1739-1742; Jun
1977
Burgaleda C. and Golde D.W. Cancer Research; 37: 1739-1742;
Jun 1977
Increased resistance to Staphylococcus aureus
infection and enhancement in serum lysozyme
activity by glucan.
Aumento dovuto al glucano della resistenza
all’infezione
da
Staphylococcus
aureus
e
dell’attività del lisozima nel siero.
Kokoshis PL; Williams DL; Cook JA; Di Luzio NR, Science 1978 Mar
24;199(4335):1340-2 (ISSN: 0036-8075)
Kokoshis PL; Williams DL; Cook JA; Di Luzio NR, Science 1978
Mar 24;199(4335):1340-2 (ISSN: 0036-8075)
Glucan, a potent reticuloendothelial stimulant, is a glucopyranose
polysaccharide derived from zymosan. Because of glucan's potential
as an immunotherapeutic agent, we performed studies in order to
determine its effect on granulopoiesis and macrophage production
in mice. One week after the i.p. injection of 4 mg of glucan, there
was a tenfold increase in colony-forming cells in the spleen and
approximately a twofold increment of cells in the bone marrow and
the peritoneal cavity capable of colony formation in vitro. There was
a relative and absolute increase in the number of
pure macrophage colonies from bone marrow and spleen. The total
macrophage content in spleen, peritoneal cavity, and bone marrow
as also increased in the treated mice. Serum from glucan-injected
mice had high colony-stimulating activity levels, and the peritoneal
macrophages elaborated increased colony-stimulating activity in
vitro as compared to controls. Peripheral white blood cell counts
were two times greater than those of control in the glucan-treated
mice. These studies indicate that glucan administration results in
increased granulocyte and macrophage production. The enhanced
leukopoiesis is probably mediated in part by augmented release of
colony-stimulating activity from macrophages. These observations
suggest that the use of glucan as an immunotherapeutic agent can
result in an increased number of available effector cells.
e
la
Il glucano è un potente stimolante reticoloendoteliale, e questo è
dimostrato, tra gli altri, anche attraverso questo studio che ha
come obiettivo di determinare gli effetti del glucano sulla
produzione di macrofagi e la granulocitopoiesi.
A seguito di un’iniezione intra peritoneale giornaliera per una
settimana di 4mg di glucano nel topo, è stato rilevato un aumento
di circa 10 volte del numero delle cellule staminali nella milza e,
un incremento di circa il doppio dei granulociti nel midollo osseo e
nella cavità peritoneale.
Il siero dei topi trattati con glucano ha presentato alti livelli di
leucociti circolanti e peritoneali degli animali trattati erano di
dimensioni doppie rispetto ai controlli.
Glucan is a potent reticuloendothelial stimulant whose
immunobiological activity is mediated, in part, by an increase in the
number and function of macrophages. In studying the role of
glucan as a mediator of antibacterial activity, we attempted to
ascertain the ability of glucan to modify the mortality of mice with
experimentally induced Gram-positive bacteremia, and to enhance
antibacterial defenses in rats as denoted by serum lysozyme and
phagocytic activity. After intravenous administration of glucan,
serum lysozyme concentrations were increased approximately
sevenfold over control concentrations. The increase in serum
lysozyme appeared to parallel the glucan-induced increase in
phagocytosis and induced hyperplasia of macrophages. Prior
treatment of mice with glucan significantly enhanced their survival
when they were challenged systemically with
Staphylococcus aureus. These studies indicate that glucan confers
an enhanced state of host defense against bacterial infections.
Il glucano è un potente stimolatore reticolo-endoteliale la cui
attività immunobiologica è mediata, in parte, da un aumento nel
numero e nella funzionalità dei macrofagi. Nell’ambito dello
studio del ruolo del glucano come mediatore dell’attività
antibatterica, abbiamo tentato di accertare la capacità del
glucano di modificare la mortalità in topi con una batteremia
gram-positiva indotta sperimentalmente, e di aumentare le
difese antibatteriche nei ratti, come indicato dal lisozima nel siero
e dall’attività fagocitica. Dopo la somministrazione endovenosa di
glucano, le concentrazioni di lisozima nel siero sono state
approssimativamente di sette volte maggiori rispetto a quelle del
controllo. L’aumento di lisozima nel siero sembra seguire
parallelamente la crescita della fagocitosi indotta dal glucano e
l’iperplasia indotta nei macrofagi. Il trattamento preventivo dei
topi con il glucano ha migliorato significativamente la loro
sopravvivenza quando erano infettati sistemicamente con
Staphylococcus aureus. Questi studi indicano che il glucano
conferisce un miglioramento della difesa contro le infezioni
batteriche.
Immune recognition. A new receptor for beta-glucans.
Riconoscimento immunitario. Un nuovo recettore
per i beta-glucani.
Brown GD; Gordon S Sir William Dunn, School of Pathology,
University of Oxford, South Parks Road, Oxford OX1 3RE, UK..Nature
2001 Sep 6;413(6851):36-7
Brown GD; Gordon S Sir William Dunn, School of Pathology,
University of Oxford, South Parks Road, Oxford OX1 3RE,
UK..Nature 2001 Sep 6;413(6851):36-7
The carbohydrate polymers known as beta-1,3-d-glucans exert
potent effects on the immune system - stimulating antitumour and
antimicrobial activity, for example - by binding to receptors on
macrophages and other white blood cells and activating them.
Although beta-glucans are known to bind to receptors, such as
complement receptor 3 (ref. 1), there is evidence that another betaglucan receptor is present on macrophages. Here we identify this
unknown receptor as dectin-1 (ref. 2), a finding that provides new
insights into the innate immune recognition of beta-glucans.
I polimeri carbossilati noti come beta-1,3-d-glucani esercitano
potenti effetti sul sistema immunitario – ad esempio, stimolando
l’attività antitumorale ed antimicrobica – legandosi a recettori sui
macrofagi e su altri globuli bianchi del sangue ed attivandoli.
Benchè sia noto che i beta-glucani si legano a dei recettori, come
ad esempio il recettore di tipo 3 del complemento (rif. 1), vi è
evidenza della presenza di un altro recettore per il beta-glucano sui
macrofagi. Qui noi identifichiamo tale recettore sconosciuto come
dectina-1 (rif. 2), un risultato che fornisce nuovi approfondimenti
sul riconoscimento immunitario innato dei beta-glucani.
4
Immunomodulatory activities of oat beta-glucan in vitro
and in vivo.
Attività immunomodulatorie del beta-glucano dell’avena in
vitro ed in vivo.
Estrada A; Yun CH; Van Kessel A; Li B; Hauta S; Laarveld B
Animal Biotechnology Centre, Department of Animal and Poultry
Science, University of Saskatchewan, Saskatoon, Canada.
Microbiol Immunol 1997;41(12):991-8 (ISSN: 0385-5600)
Estrada A; Yun CH; Van Kessel A; Li B; Hauta S; Laarveld B Animal
Biotechnology Centre, Department of Animal and Poultry Science,
University of Saskatchewan, Saskatoon, Canada.
Microbiol Immunol 1997;41(12):991-8 (ISSN: 0385-5600)
Previous studies have shown that beta-glucans extracted from
yeast or fungi potentiate immune responses. In the present
study, the immunomodulatory activities of beta-(1-->3,1-->4)glucan, derived from oats, were investigated. The ability of oat
beta-glucan (ObetaG) to stimulate IL-1 and TNF-alpha release
from murine peritoneal macrophages and the murine
macrophage cell line P338D1, was assessed. In vitro stimulation
of macrophages with ObetaG resulted in the production of IL-1 in
a dose and time-dependent manner, whereas only small amounts
of TNF-alpha could be detected in the culture supernatants.
ObetaG also induced the production of IL-2, IFN-gamma and IL-4
secretion in a dose-dependent manner in cultured spleen cells.
The intraperitoneal administration of ObetaG in mice resulted in
the accumulation of leucocytes, predominantly macrophages, in
the peritoneal cavity. Furthermore, ObetaG was tested for its
ability to enhance non-specific resistance to a bacterial challenge
in mice. Survival of mice challenged with Staphylococcus aureus
was enhanced by a single intraperitoneal administration of 500
microg of ObetaG 3 days prior to bacterial challenge. In
conclusion, these studies demonstrated that ObetaG possesses
immunomodulatory activities capable of stimulating immune
functions both in vitro and in vivo.
Precedenti studi hanno mostrato che i beta-glucani estratti dai lieviti
o dai funghi potenziano le risposte immunitarie. Nel presente studio
sono state investigate le attività immunomodulatorie del beta-(1-->3,
1-->4)-glucano, derivato dall’avena. È stata valutata la capacità del
beta-glucano dell’avena (ObetaG) di stimolare il rilascio di IL-1 e
TNF-alfa da macrofagi peritoneali murini e da macrofagi della linea
cellulare murina P388D1. La stimolazione in vitro dei macrofagi con
ObetaG ha causato la produzione di IL-1 in modo dose- e tempodipendente, mentre solamente piccole quantità di TNF-alfa sono
state rinvenute nei surnatanti della cultura. ObetaG ha inoltre indotto
la produzione di IL-2, IFN-gamma, e la secrezione di IL-4 in modo
dose-dipendente in cellule coltivate della milza. La somministrazione
intraperitoneale di ObetaG nei topi ha causato l’accumulo di leucociti,
prevalentemente di macrofagi, nella cavità peritoneale. Inoltre, è
stata esaminata la capacità di ObetaG di migliorare la resistenza
aspecifica ad un’aggressione batterica nei topi. La sopravvivenza di
topi infettati con Staphylococcus aureus è stata migliorata da
un’unica somministrazione intraperitoneale di 500 microgrammi di
ObetaG 3 giorni prima dell’infezione. In conclusione, questi studi
dimostrano che ObetaG possiede attività immunomodulatorie tali da
stimolare le funzioni immunitarie sia in vitro che in vivo.
Macrophage activation in vitro by chemically cross-linked
(1----3)-beta-D-glucans.
Attivazione dei macrofagi in vitro tramite (1----3)-beta-Dglucani sottoposti a crosslink chimico.
Adachi Y; Ohno N; Ohsawa M; Oikawa S; Yadomae T Tokyo
Chem Pharm Bull (Tokyo) 1990 Apr;38(4):988-92 College of
Pharmacy, Japan.
Adachi Y; Ohno N; Ohsawa M; Oikawa S; Yadomae T Tokyo Chem
Pharm Bull (Tokyo) 1990 Apr;38(4):988-92 College of Pharmacy,
Japan.
The chemical cross-linking of soluble (1----3)-beta-D-glucans
having molecular weights of 21000 (CL 3 h) and 6400 (CL 6 h),
and laminarin (CL LAMI), which showed negligible biological
activity, by epichlorohydrin provided rigid particles. These
particles showed no gel-to-sol transition upon the addition of
sodium hydroxide. We compared the effects of chemical crosslinking on the biological activities of glucans. The alternative
complement pathway was not activated by any of the crosslinked glucans. Glucose consumption, lysosomal enzyme release,
and interleukin-1 production by mouse resident peritoneal
macrophages incubated in vitro were strongly induced by CL 3 h,
CL 6 h and CL LAMI. However, cross-linked dextran, Sephadex,
did not exhibit any of these biological activities. These results
suggested that chemical cross-linking of (1----3)-beta-D-glucans
enhances macrophage activities without opsonization by
complement components.
Il crosslink chimico degli (1----3)-beta-D-Glucani di peso molecolare
21000 (CL 3 h) e 6400 (CL 6 h), e della laminarina (CL LAMI), che
ha mostrato un’attività biologica trascurabile, con epicloroidrina ha
fornito particelle rigide. Tali particelle non hanno mostrato
transizione gel-sol a seguito dell’aggiunta di idrossido di sodio.
Abbiamo confrontato gli effetti del crosslink chimico sull’attività
biologica dei glucani. La via alternativa del complemento non è
stata attivata da nessuno dei glucani sottoposti a crosslink. CL 3 h,
CL 6 h e CL LAMI hanno fortemente indotto: il consumo di glucosio,
il rilascio di enzimi lisosomiali e la produzione di interleuchina-1 da
parte di macrofagi residenti nel peritoneo di topi e incubati in vitro.
Peraltro, il destrano sottoposto a crosslink, il Sephadex, non ha
manifestato nessuna di queste attività biologiche. Questi risultati
suggeriscono che il crosslink chimico degli (1----3)-beta-D-glucani
accresce le attività macrofagiche senza opsonizzazione da parte di
componenti complementari.
5
Effects of fungal beta-glucan and interferon-gamma on
the secretory functions of murine alveolar macrophages.
Effetti di beta-glucano ed interferone gamma derivato da
funghi sulle funzioni secretorie dei macrofagi alveolari
murini.
J Leukoc Biol 1996 Jul;60(1):118-24 (ISSN: 0741-5400)
Sakurai
T;
Ohno
N;
Yadomae
T
Laboratory of Immunopharmacology of Microbial Products,
School of Pharmacy, Tokyo University of Pharmacy and Life
Science, Japan.
We investigated the effect of a fungal component, soluble betaglucan, on secretory functions of murine alveolar macrophages
(AMs) in vitro. Stimulation by beta-glucan (500 microg/mL) or
interferon-gamma (IFN-gamma; 100 U/mL) alone had a slight
effect on AM functions, but when AMs were incubated together
with beta-glucan and IFN-gamma, the production and secretion
of some immune mediators, such as nitric oxide, interleukin-1
(IL-1), IL-6, and tumor necrosis factor-alpha (TNF-alpha), were
markedly augmented. This combined effect of beta-glucan and
IFN-gamma was based on a priming effect of IFN-gamma,
because prestimulation with IFN-gamma followed by beta-glucan
induced high nitric oxide production of AMs, but reversal of the
sequence of treatments had only a slight effect. We also found
that preincubation of AMs with IFN-gamma enhanced the binding
of fluorescein-labeled beta-glucan on the AM surface, and this
increased binding was abrogated to the control level by the
addition of three species of soluble unlabeled (1-->3)-beta-Dglucans but not by soluble alpha-glucan. These data imply that
the priming effect of IFN-gamma on the AM response to betaglucan was dependent, at least in part, on the enhancement of
beta-glucan specific binding sites on the AM surface. It was
suggested that IFN-gamma is one of the principal factors
controlling the pulmonary immune system against both severe
fungal infection and inflammation via AM activation at the alveoli.
J Leukoc Biol 1996 Jul;60(1):118-24 (ISSN:0741-5400)
Sakurai T; Ohno N; Yadomae T
Laboratory of Immunopharmacology of Microbial Products, School
of Pharmacy,
Tokyo University of Pharmacy and Life Science, Japan.
Abbiamo investigato l’effetto di un componente fungino, il betaglucano solubile, sulle funzioni secretorie di macrofagi alveolari
murini (AM) in vitro. La stimolazione con solo beta-glucano (500
microg/mL) o solo interferone gamma (IFN-gamma, 100 U/mL) ha
avuto un lieve effetto sulle funzioni degli AM, ma si è osservato un
marcato aumento nella produzione e secrezione di alcuni mediatori
della risposta immunitaria, come l’ossido di azoto, l’interleuchina-1
(IL-1), l’IL-6 e il fattore alfa di necrosi tumorale (TNF-alfa) quando
gli AM sono stati incubati contemporaneamente con beta-glucano e
IFN-gamma. Questo effetto combinato del beta-glucano e dell’IFNgamma è basato su un effetto di innesco dell’IFN-gamma, dato che
la prestimolazione con IFN-gamma seguita dal beta-glucano ha
causato un’elevata produzione di ossido di azoto da parte degli AM,
mentre il ribaltamento della sequenza di trattamento ha avuto solo
un lieve effetto. Abbiamo inoltre scoperto che la preincubazione
degli AM con IFN-gamma ha aumentato il legame sulla superficie
degli AM con il beta-glucano marcato con fluorescina, e che questo
aumento di legame è stato ridotto fino al livello dei controlli
aggiungendo tre specie di (1-->3)-beta-D-glucani solubili non
marcati, mentre non è stato ridotto aggiungendo alfa-glucani
solubili. Questi dati implicano che l’effetto di innesco dell’IFNgamma sulla risposta degli AM ai beta-glucani dipende, almeno in
parte dall’aumento di siti di legame specifici per il beta-glucano
sulla superficie degli AM. È stato suggerito che l’IFN-gamma sia uno
dei fattori principali nel controllo del sistema immunitario
polmonare nei confronti di gravi infezioni ed infiammazioni fungine
tramite l’attivazione degli AM negli alveoli.
Intravenously administered (1----3)-beta-D-glucan, SSG,
obtained from Sclerotinia sclerotiorum IFO 9395
augments murine peritoneal macrophage functions in
vivo.
La somministrazione endovenosa di (1----3)-beta-Dglucano, SSG, ottenuto dalla Sclerotinia sclerotiorum
IFO9395 incrementa la funzionalità dei macrofagi
peritoneali murini in vivo.
Effect of intravenously (i.v.) or intraperitoneally (i.p.)
administered (1----3)-beta-D-glucan, SSG, obtained from
Sclerotinia sclerotiorum IFO 9395 on the murine peritoneal
macrophage (PM) functions were examined. A single i.v.
administration of SSG increased the number of PMs at a dose of
250 micrograms/mouse, and the peak appeared 4 d after
administration. However, no special change was observed on
peritoneal exude cell (PEC) populations. These PMs showed
augmented lysosomal enzyme activity and the peaks appeared in
2 phases, on days 2 and 10. In contrast, SSG administered i.p.
(250 micrograms/mouse) increased the number of PMs and
enhanced the lysosomal enzyme activity of PMs from day 4, and
a broad peak appeared until days 8--12. The populations of PECs
were also changed by i.p. injection of SSG. Additionally, SSG
administered i.v. enhanced phagocytic activity, H2O2 production
and interleukin 1 (IL-1) production, and the kinetics of the
activation differed depending on the activities. These data
suggest that the effects of SSG on macrophage functions are
different depending on administration routes, and there are some
different mechanisms in the activation of macrophages in vivo by
SSG.
Sono stati esaminati gli effetti della somministrazione
endovenosa
(i.v.) o intraperitoneale (i.p.) di (1----3)-beta-D-glucano, SSG,
ottenuto dalla Sclerotinia sclerotiorum IFO 9395 sulla
funzionalità dei macrofagi peritoneali murini (PM). Una singola
somministrazione i.v. di SSG ad una dose di 250
microgrammi/topo ha accresciuto il numero di PM, ed il picco è
comparso 4 gg dopo la somministrazione. Comunque, non si è
osservato nessun particolare cambiamento nelle popolazioni
delle cellule dell’essudato peritoneale (PEC). Questi PM hanno
mostrato un incrementata attività dell’enzima lisosomiale ed i
relativi picchi sono comparsi in 2 fasi, al giorno 2 ed al giorno
10. Per contro, la somministrazione i.p. di SSG (250
microgrammi/topo) ha accresciuto il numero dei PM ed
incrementato l’attività dell’enzima lisosomiale dei PM a partire dal
giorno 4, ed è comparso un ampio picco fino ai giorni 8--12.
Anche le popolazioni di PEC sono state modificate dall’iniezione
i.p. di SSG. Inoltre, l’SSG somministrato i.v. ha aumentato
l’attività fagocitica, e la produzione di H202 e di interleuchina 1
(IL-1), inoltre la cinetica di attivazione è risultata differente in
dipendenza dalle attività. Questi dati suggeriscono che gli effetti
dell’SSG sulla funzionalità dei macrofagi differiscono in
dipendenza dalle vie di somministrazione, e che esistono
differenti meccanismi nell’attivazione dei macrofagi in vivo da
parte dell’SSG.
Sakurai
T;
Ohno
N;
Yadomae
T,Laboratory
of
Immunopharmacology of Microbial Products, Tokyo College of
Pharmacy, Japan. Chem Pharm Bull (Tokyo) 1992
Aug;40(8):2120-4 (ISSN:0009-2363)
Sakurai
T;
Ohno
N;
Yadomae
T,Laboratory
of
Immunopharmacology of Microbial Products, Tokyo College of
Pharmacy, Japan. Chem Pharm Bull (Tokyo) 1992 Aug;40(8):21204 (ISSN:0009-2363)
6
PGG-glucan, a soluble beta-(1,3)-glucan, enhances the
oxidative burst response, microbicidal activity, and
activates an NF-kappa B-like factor in human PMN:
evidence for a glycosphingolipid beta-(1,3)-glucan
receptor.
Il PGG-glucano, un beta-(1,3)-glucano solubile, migliora la
risposta di attivazione metabolica, l’attività microbicida ed
attiva un fattore NF-kappa B-simile nei PMN umani:
evidenza di un recettore glicosfingolipidico per il beta-(1,3)glucano.
PGG-Glucan, a soluble beta-(1,6)-branched beta-(1,3)-linked
glucose homopolymer derived from the cell wall of the yeast
Saccharomyces cerevisiae, is an immunomodulator which
enhances leukocyte anti-infective activity and enhances myeloid
and megakaryocyte progenitor proliferation. Incubation of human
whole blood with PGG-Glucan significantly enhanced the oxidative
burst response of subsequently isolated blood leukocytes to both
soluble and particulate activators in a dose-dependent manner,
and increased leukocyte microbicidal activity. No evidence for
inflammatory cytokine production was obtained under these
conditions. Electrophoretic mobility shift assays demonstrated that
PGG-Glucan induced the activation of an NF-kappaB-like nuclear
transcription factor in purified human neutrophils. The binding of
3H-PGG-Glucan to human leukocyte membranes was specific,
concentration-dependent, saturable, and high affinity (Kd
approximately 6 nM). A monoclonal antibody specific to the
glycosphingolipid lactosylceramide was able to inhibit activation of
the NF-kappaB-like factor by PGG-Glucan, and ligand binding data,
including polysaccharide specificity, suggested that the PGGGlucan binding moiety was lactosylceramide. These results
indicate that PGG-Glucan enhances neutrophil anti-microbial
functions and that interaction between this beta-glucan and
human neutrophils is mediated by the glycosphingolipid
lactosylceramide present at the cell surface.
Il PGG-glucano, un omopolimero del glucosio beta-(1,6)-ramificato e
beta-(1,3)-legato derivato dalla parete cellulare del lievito
Saccaromyces cerevisiae, è un immunomodulatore che incrementa
l’attività anti-infettiva dei leucociti e la proliferazione dei precursori
mieloidi e megacariocitici. L’incubazione di sangue intero umano con
PGG-glucano ha incrementato significativamente e in modo dosedipendente la risposta di attivazione metabolica dei leucociti del
sangue successivamente isolati agli attivatori, sia solubili che
particellati, ed ha accresciuto l’attività microbicida dei leucociti. In
queste condizioni, non si è ottenuta alcuna evidenza della
produzione di citochine infiammatorie. I test di variazione della
mobilità elettroforetica hanno mostrato che il PGG-glucano induce
l’attivazione di un fattore di trascrizione nucleare NF-kappaB-simile
nei neutrofili umani purificati. Il legame del 3H-PGG-glucano alle
membrane dei leucociti umani è risultato specifico, dipendente dalla
concentrazione,
saturabile
e
ad
alta
affinità
(Kd
approssimativamente 6 nM). Un anticorpo monoclonale specifico per
il glicofosfolipide lactosilceramide è stato in grado di inibire
l’attivazione del fattore NF-kappaB-simile da parte del PGG-glucano,
ed i dati sul legame del ligando, che includono la specificità al
polisaccaride, hanno suggerito che il gruppo funzionale di legame del
PGG-glucano è il lactosilceramide. Questi risultati indicano che il
PGG-glucano incrementa le funzioni anti-microbiche dei neutrofili e
che l’interazione tra questo beta-glucano ed i neutrofili umani è
mediata dal glicosfingolipide lactosilceramide presente sulla
superficie cellulare.
Wakshull E; Brunke-reese D; Lindermuth J; Fisette L; Nathans RS;
Crowley JJ; Tufts JC; Zimmermann J; Mackin W; Adams DS
Department of Biology, Alpha-beta Technology, Worcester, MA
01605, USA Immunopharmacology 1999 Feb;41(2):89-107 (ISSN:
0162-3109)
Wakshull E; Brunke-reese D; Lindermuth J; Fisette L; Nathans RS;
Crowley JJ; Tufts JC; Zimmermann J; Mackin W; Adams DS
Department of Biology, Alpha-beta Technology, Worcester, MA
01605, USA Immunopharmacology 1999 Feb;41(2):89-107 (ISSN:
0162-3109)
7
Modulation of endotoxin- and enterotoxin-induced cytokine
release by in vivo treatment with beta-(1,6)-branched beta(1,3)-glucan.
Modulazione del rilascio di citochine mediato da endotossine
e enterotossine tramite trattamento in vivo con beta-(1,3)glucano beta-(1,6)-ramificato.
Soltys J; Quinn MT; Department of Veterinary Molecular Biology,
Montana State University, Bozeman 59717, USA.Infect Immun 1999
Jan;67(1):244-52 (ISSN: 0019-9567)
Soltys J; Quinn MT; Department of Veterinary Molecular Biology,
Montana State University, Bozeman 59717, USA.Infect Immun 1999
Jan;67(1):244-52 (ISSN: 0019-9567)
Leukocytes activated by endotoxin or enterotoxins release
proinflammatory cytokines, thereby contributing to the cascade of
events leading to septic shock. In the present studies, we analyzed
the effects of in vivo administration of a soluble immunomodulator,
beta-(1,6)-branched beta-(1,3)-glucan (soluble beta-glucan), on
toxin-stimulated cytokine production in monocytes and lymphocytes
isolated from treated mice. In vitro stimulation of lymphocytes
isolated
from
soluble
beta-glucan-treated
mice
with
lipopolysaccharide (LPS) resulted in enhanced production of
interleukin-6 (IL-6) and suppressed production of tumor necrosis
factor alpha (TNF-alpha), while stimulation of these cells with
staphylococcal enterotoxin B (SEB) or toxic shock syndrome toxin 1
(TSST-1) resulted in enhanced production of gamma interferon
(IFN-gamma) and suppressed production of IL-2 and TNF-alpha
compared to that in cells isolated from untreated mice. In vitro
stimulation of monocytes isolated from soluble beta-glucan-treated
mice with LPS also resulted in suppressed TNF-alpha production,
while stimulation of these cells with SEB or TSST-1 resulted in
suppressed IL-6 and TNF-alpha production compared to that in cells
isolated from untreated mice. Thus, the overall cytokine pattern of
leukocytes from soluble beta-glucan-treated mice reflects
suppressed production of proinflammatory cytokines, especially
TNF-alpha. Taken together, our results suggest that treatment with
soluble beta-glucan can modulate the induction cytokines during
sepsis, resulting in an overall decrease in host mortality.
I leucociti attivati dalle endotossine o dalle enterotossine rilasciano
citochine proinfiammatorie, contribuendo così alla cascata di eventi
che porta allo shock settico. Negli studi qui presentati, abbiamo
analizzato gli effetti della somministrazione in vivo di un
immunomodulatore solubile, beta-(1,3)-glucano beta-(1,6)-ramificato
(beta-glucano solubile), sulla produzione di citochine indotta dalle
tossine in monociti e linfociti isolati da topi trattati. La stimolazione in
vitro con lipopolisaccaride (LPS) di linfociti isolati da topi trattati col
beta-glucano solubile ha provocato un aumento della produzione di
interleuchina-6 (IL-6) ed una soppressione della produzione del
fattore alfa di necrosi tumorale (TNF-alfa), mentre la stimolazione di
queste cellule con enterotossina B di staffilococco (SEB) o con tossina
1 della sindrome da shock tossico (TSST-1) ha provocato un aumento
della produzione di interferone gamma (IFN-gamma) ed una
soppressione della produzione di IL-2 e TNF-alfa rispetto alle cellule
isolate da topi non trattati. Anche la stimolazione in vitro con LPS di
monociti isolati da topi trattati con beta-glucano solubile ha soppresso
la produzione di TNF-alfa, mentre la stimolazione di queste cellule con
SEB o TSST-1 ha soppresso la produzione di IL-6 e TNF-alfa rispetto
alle cellule isolate dai topi non trattati. Pertanto, lo schema
complessivo delle citochine leucocitarie provenienti da topi trattati con
glucano riflette la soppressione della produzione di citochine
proinfiammatorie, specialmente del TNF-alfa. Considerati nel
complesso, i nostri risultati suggeriscono che il trattamento con betaglucano solubile può modulare l’induzione di citochine durante la
sepsi, portando ad una diminuzione della mortalità dell’ospite.
Receptor binding and internalization of a water-soluble (1->3)-beta-D-glucan biologic response modifier in two
monocyte/macrophage cell lines.
Legame al recettore e internalizzazione di un modificatore
della risposta biologica (1-->3)-beta-D-glucano idrosolubile
in due linee cellulari di monociti/macrofagi.
Muller A; Rice PJ; Ensley HE; Coogan PS; Kalbfleish JH; Kelley JL;
Love EJ; Portera CA; Ha T; Browder IW; Williams DL
Department of Surgery, James H. Quillen College of Medicine, East
Tennessee State University, Johnson City 37614, USA. J Immunol
1996 May 1;156(9):3418-25 (ISSN: 0022-1767)
Muller A; Rice PJ; Ensley HE; Coogan PS; Kalbfleish JH; Kelley JL; Love
EJ;
Portera
CA;
Ha
T;
Browder
IW;
Williams
DL
Department of Surgery, James H. Quillen College of Medicine, East
Tennessee State University, Johnson City 37614, USA. J Immunol
1996 May 1;156(9):3418-25 (ISSN: 0022-1767)
Glucan phosphate, a water-soluble, chemically defined (1-->3)beta-D-glucan biologic response modifier, has been reported to
exert antisepsis activity and accelerate wound healing. In this study
we describe the specific binding of glucan phosphate to human and
murine monocyte/macrophage cell lines, U937 and J774A.1,
respectively. At 37 degrees C, equilibrium binding was rapidly
achieved, i.e., within 1 min. In U937 cells, binding occurred with an
affinity (Kd) of 37 microM and a Bmax of 65 x 106 binding sites/cell
at 37 degrees C. In J774A.1 cells, glucan phosphate bound with an
affinity (Kd) of 24 microM and a Bmax of 53 x 106 binding sites/cell
at 37 degrees C. In both cases there was insignificant nonspecific
binding. We further demonstrated that bound glucan phosphate
cannot be displaced by a 50-fold excess of unlabeled ligand,
suggesting internalization of glucan phosphate. Transmission
electron microscopy showed significantly increased cytoplasmic
vacuolization and significantly decreased mitotic activity in glucan
phosphate-treated U937 cells compared with that in untreated cells.
Pullulan, a random coil alpha-(1-->4)-(1-->6)-linked glucose
polymer that served as a control, did not compete for the same
binding site as glucan phosphate in either cell line, indicating the
specificity of the binding site for (1-->3)-beta-D-glucans. We
conclude that water-soluble pharmaceutical grade (1-->3)-beta-Dglucan phosphate specifically binds to and is internalized by U937
and J774A.1 cells.
È stato affermato che il glucano fosfato, un modificatore della risposta
biologica chimicamente definito come (1-->3)-beta-D-glucano
idrosolubile, presenta attività antisettica e accelera la guarigione delle
ferite. Nel presente studio, descriviamo il legame specifico del glucan
fosfato con linee cellulari umane e murine di monociti/macrofagi:
U937 e J774A.1. Il legame di equilibrio è stato raggiunto rapidamente,
cioè entro 1 min, a 37 gradi C. Nelle cellule U937, il legame si è avuto
con un’affinità (Kd) di 37 microM e con un Bmax di 65 x 10(6) siti di
legame/cellula a 37 gradi C. Nelle cellule J774A.1, il glucan fosfato si
è legato con un’affinità (Kd) di 24 microM ed un Bmax di 53 x 10(6)
siti di legame/cellula a 37 gradi C. In entrambi i casi ci sono stati
legami aspecifici non significativi. Abbiamo inoltre dimostrato che che
il glucan fosfato legato non può essere rimosso neppure da una
quantità 50 volte in eccesso di ligando non marcato, il che suggerisce
un’internalizzazione del glucan fosfato. La microscopia elettronica a
trasmissione ha mostrato una crescita significativa della
vacuolizzazione del citoplasma ed una riduzione significativa
dell’attività mitotica nelle cellule U937 trattate con il glucan fosfato,
rispetto alle medesime cellule non trattate. Il pullulano, un polimero a
catena casuale del glucosio alfa-(1-->4)-(1-->6)-legato che serviva
come controllo, non è risultato in competizione per i medesimi siti di
legame del glucan fosfato in nessuna delle due linee cellulari, il che
indica la specificità dei siti di legame per gli (1-->3)-beta-D-glucani.
Ne concludiamo che l’ (1-->3)-beta-D-glucan fosfato idrosolubile per
uso farmaceutico si lega specificamente e viene internalizzato dalle
cellule U937 e J774A.1.
8
A beta-glucan inhibitable receptor on human monocytes: its
identity with the phagocytic receptor for particulate
activators of the alternative complement pathway.
Un recettore per il beta-glucano sui monociti umani che puo’
essere inibito: sua identità con il recettore fagocitico per le
particelle attivatrici della via alternativa del complemento.
The ligand specificity of the human monocyte receptor that
mediates phagocytosis of particulate activators of the human
alternative complement pathway was defined by inhibiting the
phagocytic response with glycans known to be present in zymosan.
When monocytes in monolayers were preincubated with 100
micrograms/ml of beta-glucan and then incubated with 1.25 to 2.5
X 10(6) zymosan particles, the percentage of cells that exhibited
phagocytosis was inhibited in a time-dependent manner; maximal
inhibition occurred within 20 min of preincubation. beta-Glucan
inhibited monocyte phagocytosis of zymosan and rabbit
erythrocytes (Er) in a similar dose-dependent fashion and at 100
micrograms/ml reduced monocyte ingestion of 5 X 10(6)/ml
zymosan and 2 X 10(8)/ml Er by 63 +/- 8% and 68 +/- 16% (mean
+/- SD, n = 3), respectively. The other glycan constituent of
zymosan, mannan, was less than 1% as active, and 10 mg/ml of
mannan reduced the number of monocytes ingesting zymosan and
Er by 56 +/- 12% and 26 +/- 11%, respectively. At concentrations
as high as 500 micrograms/ml, beta-glucan had no effect on
monocyte Fc, C3b, or fibronectin receptor-mediated functions.
Enzymatic hydrolysis of beta-glucan and alpha-mannan with betaglucosidase or beta-glucanase before their incubation with
monocytes abrogated their inhibitory capacity, whereas hydrolysis
with alpha-mannosidase or alpha-glucosidase did not. Neither of the
two alpha-glucans tested (dextran T-70 and nigeran) affected
monocyte ingestion of zymosan particles or sheep erythrocytes (Es)
sensitized with rabbit 7S anti-Es (EsIgG) at concentrations as high
as 2 mg/ml. In contrast, a number of beta-glucans were active
against zymosan but not EsIgG ingestion with a 75% reduction in
the number of monocytes ingesting zymosan occurring with 100
micrograms/ml laminarin, 500 micrograms/ml soluble pachyman,
and 900 micrograms/ml of soluble pustulan. The galactan, agarose,
either in suspensions at 2 mg/ml or in a soluble portion at 600
micrograms/ml failed to affect monocyte ingestion of zymosan
particles or Er. Thus, the monocyte receptor for particulate
activators that is specifically inhibited by beta-glucan at a rate
compatible with a phagocytic process and that recognizes betaglucans but not alpha-glucans, mannan, or galactan is a betaglucan receptor.
La specificità di ligando del recettore dei monociti umani che media la
fagocitosi delle particelle attivatrici della via alternativa del
complemento è stata definita inibendo la risposta fagocitica tramite dei
glucani notoriamente presenti nello zimosano. Preincubando i monociti,
coltivati in monostrato, con 100 microgrammi/ml di beta-glucano e
successivamente incubandoli con un numero di particelle di zimosano
variabile da 1.25 a 2.5 X 10(6), si è ottenuta una riduzione tempodipendente della percentuale di cellule che esibiscono fagocitosi; la
massima inibizione si è avuta dopo 20 min di preincubazione. Il betaglucano ha inibito in maniera simile e dose-dipendente la fagocitosi
dello zimosano e degli eritrociti di coniglio (Er) e, a 100
microgrammi/ml, ha ridotto l’ingestione da parte dei monociti di 5 X
10(6) particelle/ml di zimosano e di 2 X 10(8)/ml Er, rispettivamente
del 63 +/- 8% e del 68 +/- 16% (media +/- deviazione standard, n =
3). L’altro glucano tra i costituenti dello zimosano, il mannano, era
meno dell’ 1% come composto attivo, e 10 mg/ml di mannano hanno
causato una riduzione del numero di monociti che ingeriscono
zimosano ed Er rispettivamente del 56 +/- 12% e del 26 +/- 11%. Per
concentrazioni fino a 500 microgrammi/ml, il beta-glucano non ha
avuto alcun effetto sulle funzioni mediate dai recettori Fc, C3b e per la
fibronectina. L’idrolisi enzimatica con beta-glucosidasi o beta-glucanasi
prima dell’incubazione con i monociti ha eliminato la capacità inibitoria
del beta-glucano e dell’alfa-mannano, per contro l’idrolisi con alfamannosidasi o alfa-glucosidasi non ha avuto effetto. Nessuno dei due
alfa-glucani esaminati (destrano T-70 e nigerano), fino a concentrazioni
di 2 mg/ml, ha avuto effetti sull’ingestione da parte dei monociti di
particelle di zimosano o di eritrociti di pecora (Es) sensibilizzati con 7S
anti-Es di coniglio (EsIgG). Per contro, alcuni beta-glucani si sono
dimostrati attivi nei confronti dell’ingestione di zimosano ma non
dell’ingestione di EsIgG, con una riduzione del 75% del numero di
monociti che ingeriscono zimosano che si verifica con 100
microgrammi/ml di laminarina, 500 microgrammi/ml di pachyman, e
900 microgrammi/ml di pustulan solubile. Il galattano, un agarosio, sia
in sospensione a 2 mg/ml che in soluzione a 600 microgrammi/ml non
ha avuto alcun effetto sull’ ingestione da parte dei monociti di particelle
di zimosano o di Er. Pertanto, il recettore per le particelle attivatrici dei
monociti che viene inibito specificamente dal beta-glucano ad un livello
compatibile con un processo di fagocitosi, e che riconosce i betaglucani ma non gli alfa-glucani, il mannano o il galattano, è un
recettore per il beta-glucano.
Functional beta-glucan receptor expression by a microglial
cell line
Espressione di un recettore per il beta-glucano da parte di
una linea di cellule microgliali.
In the central nervous system, the functions of microglia appear
crucial after brain damage, when phagocytes eliminate cell debris,
acting as the scavengers of the brain. Diseases where an active role
for microglia has been proposed recently include Alzheimer's
disease, the acquired immune deficiency syndrome (AIDS) and
multiple sclerosis. Only recently has it been possible to obtain a
microglial cell line retaining morphological and functional aspects of
these cells and their secretory products. Sugar receptors are
expressed by a variety of phagocytes in primary cultures, but in
contrast, are absent on the majority of the described macrophagelike cell lines. We here establish, by 4 degrees C binding
experiments, that this murine cell line, called BV-2, expresses a high
level (9.86 +/- 0.91 x 10(5); n = 3) of beta-glucan receptors. At 37
degrees C, BV-2 cells show high phagocytic power that can only be
inhibited by the free polysugar beta-laminarin (a poly-glucose) and
not by mannan (a poly-mannose) as described for macrophages.
The beta-glucan receptor expressed by the microglial cell line BV-2
is fully functional in phagocytosis of unopsonized heat-killed yeast
particles.
La funzione della microglia nel sistema nervoso centrale sembra
essere cruciale a seguito di un danno cerebrale, quando i fagociti
eliminano i detriti cellulari, agendo come gli “spazzini” del cervello. Le
patologie per le quali è stato proposto un ruolo attivo della microglia
comprendono la malattia di Alzheimer, la sindrome da
immunodeficienza acquisita (AIDS) e la sclerosi multipla. Solo di
recente è stato possibile ottenere una linea cellulare microgliale che
mantenesse gli aspetti morfologici e funzionali di questo tipo cellulare
ed i relativi prodotti di secrezione. I recettori per gli zuccheri sono
espressi da una varietà di fagociti in culture primarie ma, per contro,
sono assenti nella maggioranza delle linee cellulari macrofago-simili
già descritte. Qui noi stabiliamo, tramite esperimenti di legame a 4
gradi C, che la linea cellulare murina, denominata BV-2, esprime un
elevato livello (9.86 +/- 0.91 x 10(5); n = 3) di recettori per il betaglucano. A 37 gradi C, le cellule BV-2 mostrano un alto potere
fagocitico che può essere inibito solamente dal polisaccaride libero
beta-laminarina (un poli-glucosio) e non dal mannano (un polimannosio), in analogia a quanto descritto per i macrofagi. I recettori
per il beta-glucano espressi dalla linea cellulare microgliale BV-2 è
completamente funzionale nella fagocitosi di particelle di lievito non
opsonizzate uccise con calore.
Czop JK; Austen KF; J Immunol 1985 Apr;134(4):2588-93
0022-1767)
(ISSN:
Czop JK; Austen KF; J Immunol 1985 Apr;134(4):2588-93 (ISSN: 00221767)
Muller CD; Bocchini V; Giaimis J; Guerrieri P; Lombard Y; Poindron
P,Departement d’Immunologie, Universitè Louis Pasteur de
Strasbourg, Illkirch, France.Res Immunol 1994 May;145(4):267-75
(ISSN: 0923-2494)
Muller CD; Bocchini V; Giaimis J; Guerrieri P; Lombard Y; Poindron P,
Departement d’Immunologie, Universitè Louis Pasteur de Strasbourg,
Illkirch, France.Res Immunol 1994 May;145(4):267-75 (ISSN: 09232494)
9
Isolation of a yeast heptaglucoside that inhibits monocyte
phagocytosis of zymosan particles.
Isolamento di un eptaglucoside del lievito che inibisce la
fagocitosi da parte dei monociti di particelle di zimosano.
To isolate a unit ligand recognized by human monocyte beta-glucan
receptors, acid-solubilized oligoglucosides were prepared by partial
acid hydrolysis of purified yeast cell walls, gel filtered sequentially
on Bio-Gel P-4 and P-2, derivatized with 2-aminopyridine, and
separated by normal-phase HPLC. Ligand recognition was assessed
by quantitating the effect of pretreatment with isolated materials on
the capacities of adherent monocytes to phagocytose zymosan
particles. Partial acid hydrolysis solubilized 23 +/- 4% (mean +/SD; n = 7) of
the cell wall glucans; at an input of 50 micrograms/ml, the
solubilized products reduced the numbers of monocytes ingesting
zymosan by an average of 44%. Gel filtration of acid-solubilized
glucans on Bio-Gel P-4 revealed several peaks with phagocytosisinhibiting activity, and fractions from the peak containing the
smallest oligoglucosides, which accounted for 10 +/- 2% (mean +/SD; n = 7) of the carbohydrate applied, were pooled. Further
purification on Bio-Gel P-2 resolved this phagocytosis-inhibiting
activity to a single peak that contained apparent heptaoses and
accounted for 8 +/- 2% (mean +/- SD; n = 6) of the carbohydrate
applied. At a concentration of 0.5 microgram/ml, the
oligoglucosides pooled from the Bio-Gel P-4 and P-2 columns
reduced the numbers of ingesting monocytes by 45 +/- 1% and 42
+/- 7% (mean +/- SD; n = 3), respectively. When derivatized with
2-aminopyridine, the oligoglucosides were resolved by HPLC to a
number of peaks; a peak that eluted as an apparent heptaglucoside
contained virtually all the inhibitory activity and accounted for only
6.6 +/- 0.7% (mean +/- SD, n = 7) of the carbohydrate applied.
Gas chromatography analysis revealed only glucose and FAB-mass
spectrometric analysis showed only heptaglucoside and no
noncarbohydrate molecules. At a concentration of 1.6 ng/ml, the
derivatized yeast heptaglucoside reduced the numbers of
monocytes ingesting zymosan and glucan particles by 44 +/- 9%
(mean +/- SD; n = 5) and 45 +/- 6% (n = 3), respectively. Thus, a
heptaglucoside present in yeast cell walls is a unit ligand for human
monocyte beta-glucan receptors.
Per isolare un ligando riconosciuto dai recettori dei monociti per i
beta-glucani, sono stati preparati degli oligoglucosidi solubilizzati
con acidi tramite idrolisi acida parziale di pareti cellulari purificate
di lievito, filtrate sequenzialmente con Bio-Gel P-4 e P-2,
derivatizzate con 2-aminopiridine, e separate con HPLC
(cromatografia liquida ad alte prestazioni) in fase normale. Il
riconoscimento del ligando è stato effettuato tramite la
quantificazione dell’effetto del pretrattamento con i materiali
isolati sulle capacità dei monociti aderenti di fagocitare particelle
di zimosano. Il 23 +/- 4% (media +/- deviazione standard; n =
7) dei glucani della parete cellulare è stato solubilizzato usando
l’idrolisi acida parziale; ad un input di 50 microgrammi/ml, i
prodotti di solubilizzazione hanno ridotto in media del 44% il
numero di monociti che ingeriscono zimosano. La filtrazione su
gel di glucani solubilizzati in acidi con Bio-Gel P-4 ha evidenziato
numerosi picchi con attività inibitoria della fagocitosi, e sono state
raggruppate le frazioni del picco contenenti gli oligoglucosidi più
piccoli, che rappresentano il 10 +/- 2% (media +/- deviazione
standard; n = 7) dei carboidrati applicati. Un’ulteriore
purificazione con Bio-Gel P-2 ha permesso di risolvere tale attività
di inibizione della fagocitosi ad un singolo picco contenente eptosi
apparenti che rappresenta l’ 8+/- 2% (media +/- deviazione
standard; n = 6) dei carboidrati applicati. Gli oligoglucosidi
raccolti dalle colonne Bio-Gel P-4 e P-2 hanno ridotto il numero di
monociti che ingeriscono zimosano rispettivamente del 45 +/1% e del 42 +/- 7% (media +/- deviazione standard; n = 3). Gli
oligoglucosidi, una volta derivatizzati con 2-aminopiridina, sono
stati risoluti tramite HPLC ad alcuni picchi; uno di essi, che
rilasciava come un eptaglucoside apparente, conteneva
sostanzialmente tutta l’attività inibitoria e rappresentava
solamente il 6.6 +/- 0.7% (media +/- deviazione standard; n =
7) dei carboidrati applicati. L’analisi gas-cromatografica ha
evidenziato solamente glucosio e l’analisi spettrometrica FABmass ha mostrato solo eptaglucoside e nessuna molecola di
elementi diversi dai carboidrati. L’eptaglucoside derivatizzato dal
lievito, alla concentrazione di 1.6 ng/ml, ha ridotto del 44 +/- 9%
(media +/- deviazione standard; n = 5) e del 45 +/- 6% (n = 3)
il numero di monociti che ingeriscono rispettivamente zimosano e
particelle di glucano. Pertanto, uno degli eptaglucosidi presenti
sulla parete cellulare dei lieviti è un unità ligando per i recettori
del beta-glucano dei monociti umani.
Janusz MJ; Austen KF; Czop JK, Department of Medicine, Harvard
Medical School, Boston, MA J Immunol 1989 Feb 1;142(3):95965 (ISSN: 0022-1767).
Janusz MJ; Austen KF; Czop JK, Department of Medicine,
Harvard Medical School, Boston, MA J Immunol 1989 Feb
1;142(3):959-65 (ISSN: 0022-1767).
10
Isolation of soluble yeast beta-glucans that inhibit human
monocyte phagocytosis mediated by beta-glucan receptors.
Janusz MJ; Austen KF; Czop JK J
15;137(10):3270-6 (ISSN: 0022-1767)
Immunol
1986
Nov
The trypsin-sensitive receptor that mediates phagocytosis of
unopsonized zymosan particles by human monocytes has been
designated as a beta-glucan receptor because of its functional
inhibition by specific algal and plant beta-glucans. Soluble ligands
that are chemically and structurally identical to beta-glucan
constituents of zymosan were isolated from a carbohydrateenriched fraction of yeast extract by sequential chromatography on
DE-cellulose, SP-Sephadex, and Con A-Sepharose. Preincubation of
adherent human monocytes with 278, 210, and 2.5 micrograms/ml
hexose equivalents in pooled chromatographic fractions from DEcellulose, SP-Sephadex, and Con A-Sepharose, respectively,
effected 50% reductions in subsequent phagocytosis of zymosan
particles without affecting Fc-mediated ingestion of IgG-coated
sheep erythrocytes (ESIgG). The purified yeast extract-derived
beta-glucans, which contained 92% glucose and 8% mannose by
gas chromatographic analysis and eluted from a Sephacryl S-200
column as a broad peak with a Kav of 0.39 and estimated molecular
sizes of from 20,000 to 70,000 m.w., required only 3.5 +/- 0.9
micrograms/ml (mean +/- SD, n = 6), as compared with 31.5
micrograms/ml of the algal beta-glucan laminarin to achieve 50%
decreases in zymosan ingestion. Alternatively, soluble yeast betaglucans with estimated molecular sizes of from 2 X 10(5) to 2 X
10(6) were prepared from yeast glucan particles, which contained
98% glucose and 0% mannose, by sonication and sequential
centrifugation at 15,000 and 100,000 X G for 30 and 60 min,
respectively. Monocyte ingestion of zymosan was reduced by 50%
by pretreatment with 60 ng/ml of the soluble beta-glucans in
15,000 X G supernatants, whereas ingestion of ESIgG was
unaffected by as much as 50 micrograms/ml of this material. Partial
acid hydrolysis of soluble glucan-derived beta-glucans in 15,000 X G
supernatants followed by gel filtration on Bio-Gel P-4 revealed two
well-defined peaks within the inclusion volume of the column with
phagocytosis-inhibiting activity. Oligoglucosides that eluted at a Kav
of 0.46 had an estimated molecular size of 2,000 m.w. and effected
a 48% reduction in zymosan ingestion at inputs of 2 to 5
micrograms/ml, and smaller oligoglucosides with a Kav of 0.82 and
an estimated molecular size of 1,000 m.w. effected a 50%
reduction at inputs of 25 micrograms/ml. Preincubation of
monocytes for 2 min with 25 micrograms/ml of the oligoglucosides
with estimated molecular size of 1,000 m.w. and with 50 ng/ml of
soluble glucan-derived beta-glucans in 100,000 X G supernatants
reduced zymosan ingestion by 41% +/- 4 and 44% +/- 3 (mean
+/- SD, n = 3), respectively.
Isolamento di beta-glucani solubili del lievito che
inibiscono la fagocitosi dei monociti umani mediata dai
recettori per il beta-glucano.
Janusz MJ; Austen KF; Czop JK
15;137(10):3270-6 (ISSN: 0022-1767)
J
Immunol
1986
Nov
Il recettore sensibile alla tripsina, che media la fagocitosi di
particelle non opsonizzate di zimosano da parte dei monociti
umani, è stato classificato come recettore per il beta-glucano a
causa della sua inibizione funzionale da parte di specifici betaglucani ricavati dalle alghe e dalle piante. A partire da una frazione
di estratto di lievito arricchita in carboidrati, utilizzando la
cromatografia sequenziale su DE-cellulosa, SP-sephadex e Con ASepharose, sono stati isolati dei ligandi solubili chimicamente e
strutturalmente identici ai beta-glucani che costituiscono lo
zimosano. La preincubazione di monociti umani aderenti con 278,
210 e 2.5 microgrammi/litro di esoso equivalenti in frazioni
cromatografiche raggruppate da DE-cellulosa, SP-sephadex e Con
A-Sepharose, rispettivamente, ha ridotto del 50% la successiva
fagocitosi delle particelle di zimosano, senza effetto sull’ingestione
mediata dagli Fc di eritrociti di pecora rivestiti con IgG (ESIgG). I
beta-glucani derivati dall’estratto purificato di lievito, contenente il
92% di glucosio e l’8% di mannosio secondo l’analisi gas
cromatografica ed eluito da una colonna Sephacryl S-200 come un
picco ampio con una Kav di 0.39 e delle dimensioni molecolari
stimate da 20000 a 70000 m.w., hanno richiesto solo 3.5 +/- 0.9
microgrammi/ml del beta-glucano algale laminarina per causare
una diminuzione del 50% nell’ingestione di zimosano. In
alternativa, i beta-glucani solubili del lievito con dimensioni
molecolari stimate da 2 X 10(5) a 2 X 10(6) sono stati preparati da
particelle di glucano del lievito, contenenti il 98% di glucosio e lo
0% di mannosio, rispettivamente per sonicazione e
centrifugazione sequenziale a 15000 e 100000 X G per 30 e 60
min. L’ingestione dello zimosano da parte dei monociti è stata
ridotta del 50% grazie al pretrattamento con 60 ng/ml di beta
glucani solubili contenuti nei surnatanti ottenuti a 15000 X G,
mentre l’ingestione di ESIgG è risultata non influenzata da
quantità di questo materiale dell’ordine dei 50 microgrammi/ml.
L’idrolisi acida parziale dei beta glucani derivati dai glucani solubili
contenuti nei surnatanti ottenuti a 15000 X G, seguita da
filtrazione di gel su Bio-Gel P-4, ha rivelato due picchi ben definiti
all’interno del volume di inclusione della colonna che presenta
attività di inibizione fagocitotica. Gli oligoglucosidi che rilasciano ad
una Kav di 0.46 hanno una dimensione molecolare dell’ordine di
2000 m.w. ed hanno ridotto l’ingestione di zimosano del 48% per
input da 2 a 5 microgrammi/ml, mentre gli oligoglucosidi più
piccoli che rilasciano ad una Kav di 0.82 e hanno una dimensione
molecolare dell’ordine di 1000 m.w. hanno ridotto l’ingestione di
zimosano del 50% per input di 25 microgrammi/ml. La
preincubazione di monociti per 2 min con 25 microgrammi/ml degli
oligoglucosidi con dimensione molecolare dell’ordine di 1000 m.w.
e con 50 ng/ml di beta glucani derivati da glucani solubili
contenuti nei surnatanti ottenuti a 100000 X G ha ridotto
l’ingestione di zimosano rispettivamente del 41% 4 e del 44%
3 (media SD, n = 3).
11
Human monocyte scavenger receptors are pattern
recognition receptors for (1-->3)-beta-D-glucans.
Rice PJ; Kelley JL; Kogan G; Ensley HE; Kalbfleisch JH; Browder
IW; Williams DL ; Department of Pharmacology, James H.
Quillen College of Medicine, East Tennessee State University,
Building 119 Room 1-29, Dogwood Lane, Johnson City, TN
37614-1708, USA. J Leukoc Biol 2002 Jul;72(1):140-6
Glucans are cell wall constituents of fungi and bacteria that
bind to pattern recognition receptors and modulate innate
immunity, in part, by macrophage activation. We used surface
plasmon resonance to examine the binding of glucans, differing
in fine structure and charge density, to scavenger receptors on
membranes isolated from human monocyte U937 cells.
Experiments were performed at 25 degrees C using a biosensor
surface with immobilized acetylated low density lipoprotein
(AcLDL). Inhibition of the binding by polyinosinic acid, but not
polycytidylic acid, confirmed the interaction of scavenger
receptors. Competition studies showed that there are at least
two AcLDL binding sites on human U937 cells. Glucan
phosphate interacts with all sites, and the CM-glucans and
laminarin interact with a subset of sites. Polymer charge has a
dramatic effect on the affinity of glucans with macrophage
scavenger receptors. However, it is also clear that human
monocyte scavenger receptors recognize the basic glucan
structure independent of charge.
Specificity of membrane complement receptor type
three (CR3) for beta-glucans.
Ross GD; Cain JA; Myones BL; Newman SL; Lachmann PJ
Complement 1987;4(2):61-74 (ISSN: 0253-5076)
The binding of the iC3b receptor (CR3) to unopsonized
zymosan was shown to result from CR3 attachment to cell wall
beta-glucans. A specificity of neutrophil responses for betaglucan was first suggested by a comparison of yeast
(Saccharomyces cerevisiae) cell wall components for
stimulation of a neutrophil superoxide burst. Neutrophils
responded poorly to heat-killed yeast, but gave increasingly
better responses to cell wall polysaccharides devoid of proteins
(zymosan) and nearly pure beta-glucan particles derived from
zymosan. Zymosan triggered a burst that was 29% as great as
that stimulated by phorbol myristate acetate (PMA), and betaglucan particles stimulated a burst that was 72% as great as
that produced by PMA. Phagocytic responses to yeast were
also inhibited by soluble glucans but not by soluble mannans.
Three types of experiments demonstrated a role for CR3 in
these responses. First, neutrophil ingestion of either yeast or
yeast-derived beta-glucan particles was blocked by monoclonal
anti-CR3, fluid-phase iC3b, or soluble beta-glucan from barley.
Monocyte ingestion of beta-glucan particles was also blocked
by anti-CR3, but not by anti-CR1 or anti-C3. Second, the
neutrophil superoxide burst response to either zymosan or
beta-glucan particles was blocked by anti-CR3 or fluid-phase
iC3b, and was completely absent with neutrophils from 3
patients with an inherited deficiency of CR3. Third, CR3 was
isolated from solubilized neutrophils by affinity chromatography
on beta-glucan-Sepharose.
I recettori “scavenger” dei monociti umani sono
recettori di pattern recognition per gli (1-->3)-beta-Dglucani.
Rice PJ; Kelley JL; Kogan G; Ensley HE; Kalbfleisch JH; Browder
IW; Williams DL ; Department of Pharmacology, James H.
Quillen College of Medicine, East Tennessee State University,
Building 119 Room 1-29, Dogwood Lane, Johnson City, TN
37614-1708, USA. J Leukoc Biol 2002 Jul;72(1):140-6
I glucani sono costituenti della parete cellulare dei funghi e dei
batteri che si legano a recettori di pattern recognition e
modulano, in parte, l’immunità innata tramite l’attivazione dei
macrofagi. Abbiamo utilizzato la risonanza plasmonica di
superficie per esaminare il legame dei glucani, che differiscono
nella struttura fine e nella densità di carica, ai recettori
“scavenger” su membrane cellulari isolate da monociti umani
U937. Gli esperimenti sono stati condotti a 25 gradi C utilizzando
una superficie del biosensore dove è immobilizzata una
lipoproteina acetilata a bassa densita (AcLDL). L’inibizione del
legame, che avviene con l’acido polinosinico, ma non con l’acido
policitidilico, ha confermato l’interazione con i recettori
“scavenger”. Studi di confronto hanno mostrato che ci sono
almeno due siti di legame per le AcLDL sulle cellule U937 umane.
Il glucan fosfato interagisce con tutti i siti, i CM-glucani e la
laminarina interagiscono con un sottoinsieme di siti. La carica del
polimero ha un pesante effetto sull’affinità dei glucani per i
recettori “scavenger” dei macrofagi. È comunque chiaro che i
recettori “scavenger” dei monociti umani riconoscono la struttura
di base del glucano indipendentemente dalla carica.
Specificità del recettore di membrana
complemento tipo 3 (CR3) per i beta-glucani.
per
il
Ross GD; Cain JA; Myones BL; Newman SL; Lachmann PJ
Complement 1987;4(2):61-74 (ISSN: 0253-5076)
È stato dimostrato che il legame del recettore iC3B (CR3) con lo
zimosano non opsonizzato è dovuto al legarsi di CR3 coi betaglucani della parete cellulare. Una specificità della risposta dei
neutrofili al beta-glucano è stata originariamente suggerita dal
confronto tra le capacità dei componenti della parete cellulare nel
lievito (Saccaromyces cerevisiae) di stimolare un “burst” del
perossido (superoxide burst) nei neutrofili. I neutrofili hanno
risposto scarsamente al lievito ucciso al calore, ma hanno dato
risposte sempre migliori ai polisaccaridi della parete cellulare
priva di proteine (zimosano) ed alle particelle quasi pure di betaglucano derivate dallo zimosano. Lo zimosano ha indotto un
“burst” del 29% maggiore rispetto a quello indotto dal forbolo
miristato acetato (PMA), e le particelle di beta-glucano hanno
indotto un “burst” del 72 % maggiore rispetto a quello prodotto
dal PMA. Le risposte fagocitiche al lievito sono state inoltre inibite
da glucani solubili, ma non da mannani solubili. Tre tipi di
esperimento hanno dimostrato un ruolo del CR3 in queste
risposte. Primo: l’ingestione da parte dei neutrofili sia di lievito
che di particelle di beta-glucano derivate dal lievito è stata
bloccata da anti-CR3 monoclonali, iC3b in fase liquida o betaglucani solubili derivati dall’orzo. L’ingestione di particelle di betaglucano da parte dei monociti è stata inoltre bloccata dagli antiCR3, ma non dagli anti-CR1 o dagli anti-C3. Secondo: la risposta
allo zimosano o alle particelle di beta-glucano in termini di
“burst” del perossido nei neutrofili è stata bloccata dagli anti-CR3
o dagli iC3b in fase liquida, ed è stata del tutto assente nei
neutrofili provenienti da 3 pazienti con una carenza ereditaria di
CR3. Terzo: il CR3 è stato isolato da neutrofili solubilizzati tramite
cromatografia di affinità su beta-glucan-sefarosio.
12
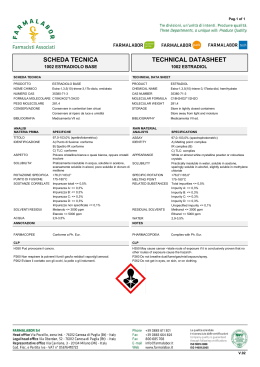
GLUCAN
Antibacterial Activity and
synergism with antibiotics
HO
O
CH 2 OCH 2 COO
-
Na
+
O
OH
HO
O
CH 2 OH
OH
13
O
A phase II multicenter, double-blind, randomized, placebocontrolled study of three dosages of an immunomodulator
(PGG-glucan) in high-risk surgical patients.
Babineau TJ; Hackford A; Kenler A; Bistrian B; Forse RA; Fairchild
PG; Heard S; Keroack M; Caushaj P; Benotti P ;Department of
Surgery, Deaconess Hospital, Harvard Medical School, Boston.
Arch Surg 1994 Nov;129(11):1204-10 (ISSN: 0004-0010)
OBJECTIVE: To examine the safety and efficacy of multiple doses of
PGG-glucan
(poly-[1-6]-B-D-glucopyranosyl-[1-3]-B-Dglucopyranose) in high-risk patients undergoing major thoracic or
abdominal surgery. DESIGN: An interventional, multicenter, doubleblind, randomized, placebo-controlled study. SETTING: Four
university-affiliated medical centers. PATIENTS: Sixty-seven highrisk patients undergoing major thoracic or abdominal surgery.
INTERVENTION: Patients were randomized in a 1:1:1:1 ratio to
receive saline placebo or PGG-glucan at a dose of 0.1 mg/kg, 0.5
mg/kg, and 1.0 mg/kg or 2.0 mg/kg. One dose was administered
before surgery and three doses were administered after surgery.
MAIN OUTCOME MEASURES: To examine the safety and efficacy of
PGG-glucan infusion and to identify potentially important factors for
a planned phase III study. RESULTS: A dose-response trend with
regard to infection incidence among patients who received PGGglucan was observed. Serious infections occurred in four patients
who received placebo and in three patients who received PGGglucan at a dose of 0.1 mg/kg. However, only one patient who
received PGG-glucan at a high dose had a serious infection. The
incidence and severity of adverse events was comparable in all
groups. CONCLUSIONS: PGG-glucan was generally safe and well
tolerated, may decrease postoperative infection rates, and warrants
further investigation in a planned phase III trial.
Uno studio di fase II multicentrico, in doppio cieco,
controllato con placebo di tre dosaggi di un
immunomodulatore (PGG-glucano) in pazienti chirurgici
ad alto rischio.
Babineau TJ; Hackford A; Kenler A; Bistrian B; Forse RA;
Fairchild PG; Heard S; Keroack M; Caushaj P; Benotti P ;
Department of Surgery, Deaconess Hospital, Harvard Medical
School, Boston.
Arch Surg 1994 Nov;129(11):1204-10 (ISSN: 0004-0010)
OBIETTIVO: esaminare la sicurezza e l’efficacia di diversi
dosaggi di PGG-glucano (poli-[1,6]-B-D-glucopiranosil-[1,3]-B-Dglucopiranosio) in pazienti ad alto rischio sottoposti a gravi
interventi chirurgici toracici o addominali. PROGETTO: uno
studio basato su interventi chirurgici, multicentrico, in doppio
cieco, randomizzato, controllato tramite placebo. AMBIENTE:
quattro centri medici affiliati ad università. PAZIENTI:
sessantasette pazienti ad alto rischio sottoposti a gravi
interventi chirurgici toracici o addominali. INTERVENTO: i
pazienti sono stati randomizzati con un rapporto 1:1:1:1 per
ricevere: soluzione salina placebo o PGG-glucano in dosi da: 0.1
mg/kg, 0.5 mg/kg, e 1.0 mg/kg o 2.0 mg/kg. Una dose è stata
somministrata prima dell’intervento e tre dosi sono state
somministrate dopo l’intervento. PRINCIPALI MISURE: l’esame
della sicurezza e dell’efficacia delle iniezioni di PGG-glucano e
l’identificazione di fattori potenzialmente importanti per uno
studio pianificato di fase III. RISULTATI: la tendenza doserisposta rispetto all’incidenza di infezioni tra i pazienti che hanno
ricevuto il PGG-glucano. Infezioni gravi si sono verificate in
quattro pazienti che hanno ricevuto il placebo ed in tre pazienti
che hanno ricevuto il PGG-glucano alla dose di 0.1 mg/kg.
Comunque, solo un paziente che ha ricevuto il PGG-glucano a
dosi elevate ha presentato un infezione grave. L’incidenza e la
gravità di eventi sfavorevoli è stata comparabile in tutti i gruppi.
CONCLUSIONI: il PGG-glucano è risultato generalmente sicuro e
ben tollerato, in grado di diminuire il tasso di infezioni postoperatorie e in grado di garantire ulteriori ricerche in uno studio
pianificato di fase III.
Comparative tumor-inhibitory and anti-bacterial activity of
soluble and particulate glucan.
Attività inibitrice di tumori e antibatterica comparata del
glucano solubile e particellato.
A soluble fraction of particulate glucan was prepared and evaluated
for its anti-tumor and anti-bacterial activity. Thin-layer
chromatographic analysis indicated that the soluble preparation was
composed of a variety of polyglucoses.
Intravenous administration of soluble or particulate glucan resulted
in significant reductions in the growth of a syngeneic anaplastic
mammary carcinoma and melanoma B16. Survival data
demonstrated that intravenous administration of soluble or
particulate glucan prolonged survival of A/J and C57BL/6J mice with
subcutaneous tumor implants. As regards to bacterial infections,
soluble and particulate glucan decreased renal necrosis in S. aureus
challenged mice as compared to control mice. Although the exact
nature of the active soluble fraction(s) of glucan remains to be
delineated, these studies demonstrate that a soluble glucan
preparation exhibits significant anti-tumor and anti-staphylococcal
activity. The active soluble fraction of particulate glucan may be
preferable to particulate glucan in view of the inherent ease of
parenteral administration.
È stata preparata una frazione solubile del glucano particellato e
ne è stata valutata l’attività antitumorale ed antibatterica.
L’analisi cromatografica in strato sottile ha indicato che la
preparazione solubile era composta da un varietà di poliglucosi.
La somministrazione endovenosa di glucano solubile o
particellato ha prodotto una significativa inibizione della crescita
di un carcinoma mammario singenico anaplastico e di un
melanoma B16. I dati di sopravvivenza hanno dimostrato che la
somministrazione endovenosa di glucano particellato o solubile
ha prolungato la sopravvivenza di topi A/J e C57BL/6J con
impianti tumorali sottocutanei. Per quanto riguarda le infezioni
batteriche, il glucano particellato e solubile hanno ridotto la
necrosi renale nei topi infettati con S. aureus in confronto ai
controlli. Benchè l’esatta natura delle frazioni solubili attive di
glucano resti da definire, questi studi dimostrano che una
preparazione solubile di glucano presenta un’attività
antitumorale e anti stafilococcica significativa. La frazione
solubile attiva può essere preferita al glucano particellato in
vista della semplicità intrinseca nella somministrazione
parenterale.
Di Luzio NR; Williams DL; McNamee RB; Edwards BF; Kitahama A
Int J Cancer 1979 Dec 15;24(6):773-9 (ISSN: 0020-7136)
Di Luzio NR; Williams DL; McNamee RB; Edwards BF; Kitahama
A Int J Cancer 1979 Dec 15;24(6):773-9 (ISSN: 0020-7136)
14
Protective immunity against Streptococcus mutans infection
in mice after intranasal immunization with the glucanbinding region of S. mutans glucosyltransferase.
Jespersgaard C, Hajishengallis G, Huang Y, Russell MW, Smith DJ,
Michalek SM Departments of Microbiology, University of Alabama at
Birmingham, Birmingham, Alabama 35294, SA. Infect Immun 1999
Dec;67(12):6543-9
Here we present the construction and characterization of a chimeric
vaccine protein combining the glucan-binding domain (GLU) of the
gtfB-encoded
water-insoluble
glucan-synthesizing
glucosyltransferase enzyme (GTF-I) from Streptococcus mutans and
thioredoxin from Escherichia coli, which increases the solubility of
coexpressed recombinant proteins and stimulates proliferation of
murine T cells. The protective potential of intranasal (i.n.)
immunization with this chimeric immunogen was compared to that
of the GLU polypeptide alone in a mouse infection model. Both
immunogens were able to induce statistically significant mucosal
(salivary and vaginal) and serum responses (P < 0.01) which were
sustained to the end of the study (experimental day 100). Following
infection with S. mutans, sham-immunized mice maintained high
levels of this cariogenic organism ( approximately 60% of the total
oral streptococci) for at least 5 weeks. In contrast, animals
immunized with the thioredoxin-GLU chimeric protein (Thio-GLU)
showed significant reduction (>85%) in S. mutans colonization after
3 weeks (P < 0.05). The animals immunized with GLU alone
required 5 weeks to demonstrate significant reduction (>50%) of S.
mutans infection (P < 0.05). Evaluation of dental caries activity at
the end of the study showed that mice immunized with either ThioGLU or GLU had significantly fewer carious lesions in the buccal
enamel or dentinal surfaces than the sham-immunized animals (P <
0.01). The protective effects against S. mutans colonization and
caries activity following i.n. immunization with GLU or Thio-GLU are
attributed to the induced salivary immunoglobulin A (IgA) anti-GLU
responses. Although in general Thio-GLU was not significantly better
than GLU alone in stimulating salivary IgA responses and in
protection against dental caries, the finding that the GLU
polypeptide alone, in the absence of any immunoenhancing agents,
is protective against disease offers a promising and safe strategy for
the development of a vaccine against caries.
Immunità protettiva contro l’infezione da Streptococcus
mutans nei topi dopo immunizzazione intranasale con il
sito di legame del glucano della glucosiltransferasi di S.
mutans.
Jespersgaard C, Hajishengallis G, Huang Y, Russell MW, Smith
DJ, Michalek SM Departments of Microbiology, University of
Alabama at Birmingham, Birmingham, Alabama 35294, SA.
Infect Immun 1999 Dec;67(12):6543-9
Presentiamo qui la preparazione e caratterizzazione di una
proteina vaccino chimerica che combina il dominio di legame per
il glucano (GLU) della glucosiltransferasi (GTF-I), l’enzima
insolubile in acqua per la sintesi del glucano, proveniente da
Streptococcus mutans, con la tioredoxina proveniente da
Escherichia Coli, la quale incrementa la solubilità delle proteine
ricombinanti coespresse e stimola la proliferazione di cellule T
murine. Il potenziale protettivo dell’immunizzazione intranasale
(i.n.) con questo immunogeno chimerico è stato confrontato con
quello del solo polipeptide GLU in un modello di infezione
murino. Entrambi gli immunogeni sono stati in grado di indurre
risposte delle mucose (salivare e vaginale) e del siero
statisticamente significative (P<0.01) che si sono mantenute
fino al termine dello studio (giorno 100 dell’esperimento). Dopo
l’infezione con S. mutans, i topi con falsa immunizzazione hanno
mantenuto per almeno 5 settimane elevati livelli di questo
microrganismo cariogeno (che rappresenta circa il 60% del
totale degli streptococchi orali). Per contro, gli animali
immunizzati con la proteina chimerica tioredoxina-GLU (ThioGLU) hanno mostrato riduzioni significative (>85%) della
colonizzazione da S. mutans dopo 3 settimane (P<0.05). Gli
animali immunizzati con il solo GLU hanno necessitato di 5
settimane per mostrare una riduzione significativa (>50%)
dell’infezione da S. mutans (P<0.05). La valutazione dell’attività
delle carie dentali al termine dello studio ha mostrato che i topi
immunizzati con Thio-GLU o GLU hanno presentato
significativamente meno lesioni cariose nello smalto dentale o
sulla superficie della dentina rispetto ai topi con falsa
immunizzazione (P<0.01). Gli effetti protettivi contro la
colonizzazione da S. mutans dopo l’immunizzazione i.n. con GLU
o Thio-GLU si possono attribuire all’induzione della risposta
dell’immunoglobulina A (IgA) anti-GLU salivare. Benchè in
generale Thio-GLU non sia stata significativamente migliore del
solo GLU nella stimolazione dell’IgA salivare e nella protezione
contro la carie, la scoperta che il polipeptide GLU da solo, in
assenza di qualsiasi agente immunostimolatore, è protettivo
verso questa malattia offre una strategia promettente e sicura
per lo sviluppo di un vaccino contro la carie.
Prophylaxis with the immunomodulator PGG glucan
enhances antibiotic efficacy in rats infected with antibioticresistant bacteria.
La profilassi con l’immunomodulatore PGG glucano
incrementa l’efficacia degli antibiotici nei ratti infettati
con batteri antibiotico-resistenti.
The emergence of multiple antibiotic-resistant microorganisms has
led to a search for alternatives to traditional therapeutic regimens.
PGG glucan is a soluble beta-glucan immunomodulator that
selectively enhances the microbicidal activities of neutrophils and
macrophages without stimulating proinflammatory cytokine
production. In the present studies, we examined the ability of PGG
glucan to act in concert with antibiotics to decrease mortality in a
rat model of intraabdominal sepsis using antibiotic-resistant bacteria
as infectious inocula. Results of these studies demonstrated that
prophylaxis with PGG glucan in combination with antibiotics
provided enhanced protection against lethal challenge with
Esherichia coli or Staphylococcus aureus as compared with the use
of antibiotics alone.
L’emergere di molti microorganismi antibiotico-resistenti ha
portato a ricercare delle alternative ai regimi terapeutici
tradizionali.
Il
PGG
glucano
è
un
beta-glucano
immunomodulatore solubile che aumenta selettivamente le
attività microbicide dei neutrofili e dei macrofagi senza stimolare
la produzione di citochine proinfiammatorie. Nel presente studio
abbiamo esaminato la capacità del PGG glucano di agire di
concerto con gli antibiotici nel decrescere la mortalità in un
modello murino di sepsi intraaddominale ottenuto utilizzando
batteri antibiotico-resistenti come infettanti. I risultati di questi
studi hanno dimostrato che la profilassi con PGG glucano in
associazione con antibiotici ha fornito un’incrementata
protezione contro le infezioni letali da E. coli o da S. aureus,
rispetto all’uso del solo antibiotico.
Tzianabos AO; Cisneros RL ;Channing Laboratory Brigham and
Women's Hospital, Harvard Medical School Boston, Massachusetts
02115, USA. Ann N Y Acad Sci 1996 Oct 25;797:285-7 (ISSN:
0077-8923)
Tzianabos AO; Cisneros RL ;Channing Laboratory Brigham and
Women's Hospital, Harvard Medical School Boston,
Massachusetts 02115, USA. Ann N Y Acad Sci 1996 Oct
25;797:285-7 (ISSN: 0077-8923)
15
Enhanced clearance of a multiple antibiotic resistant
Staphylococcus aureus in rats treated with PGG-glucan
is associated with increased leukocyte counts and
increased neutrophil oxidative burst activity.
L’aumento dell’eliminazione di uno Staphylococcus aureus
con resistenza multipla agli antibiotici in ratti trattati con
PGG-glucano è associata con l’aumento della conta
leucocitaria e dell’attivazione metabolica dei neutrofili.
Liang J; Melican D; Cafro L; Palace G; Fisette L; Armstrong R;
Patchen ML;Alpha-Beta Technology, Inc., Worcester, MA
10605, USA.Int J Immunopharmacol 1998 Nov;20(11):595614 (ISSN: 0192-0561)
Liang J; Melican D; Cafro L; Palace G; Fisette L; Armstrong R;
Patchen ML;Alpha-Beta Technology, Inc., Worcester, MA 10605,
USA.Int J Immunopharmacol 1998 Nov;20(11):595-614 (ISSN:
0192-0561)
PGG-Glucan [Betafectin], a highly purified soluble beta-(1-6)branched
beta-(1
3)-linked
glucan
isolated
from
Saccharomyces cerevisiae, has broad in vitro and in vivo antiinfective activities unrelated to cytokine induction. Here we
present in vivo results on the anti-infective activity of PGGGlucan against a multiple antibiotic resistant Staphylococcus
aureus. PGG-Glucan (0.25-4 mg/kg) was administered
intramuscularly to male Wistar rats 48 h, 24 h, and 4 h before
and 4 h after intraperitoneal implantation of a gelatin capsule
containing 10(8)S. aureus colony forming units (CFU). Blood
samples were collected at various times after challenge to
determine CFU levels, leukocyte counts and neutrophil
oxidative burst activity; serum TNF-alpha, and IL-1beta levels
were also evaluated. The 0.25 mg/kg PGG-Glucan dose had no
effect on reducing blood CFU levels; however, PGG-Glucan
doses of 0.5 mg/kg, 1 mg/kg, 2 mg/kg or 4 mg/kg significantly
reduced blood CFU levels by 48 h after challenge. Reduced
CFU levels correlated with significantly elevated absolute
monocyte counts, absolute neutrophil counts, and neutrophil
oxidative burst activity in the absence of any effect on TNFalpha or on IL-1beta levels. In additional studies, effects on
mortality and blood CFU levels were evaluated in rats treated
with ampicillin (an antibiotic to which the S. aureus was
resistant), PGG-Glucan, or both agents. Mortality and blood
CFU levels were reduced most in combination-treated rats
compared to saline control rats or rats treated with either
ampicillin alone or PGG-Glucan alone. We conclude that in vivo
(1) PGG-Glucan can enhance clearance of an antibiotic
resistant S. aureus, (2) that this clearance is accompanied by
an increase in monocytes and neutrophils as well as a
potentiation of neutrophil oxidative microbiocidal activity
without alteration of the proinflammatory cytokine response,
and (3) PGG-Glucan can enhance the effectiveness of
traditional antibiotic treatment.
Il PGG-glucano [betafectina], un glucano solubile altamente
purificato beta-(1-6)-ramificato beta-(1-3)-legato isolato da
Saccaromyces cerevisiae, ha un’ampia attività anti-infettiva in vitro
ed in vivo, non correlata all’induzione di citochine. Qui presentiamo i
risultati dello studio dell’attività anti-infettiva del PGG-glucano verso
uno Staphylococcus aureus con resistenza multipla agli antibiotici. Il
PGG-glucano è stato somministrato (0.25-4 mg/kg) per via
intramuscolare a ratti Wistar maschi 48 ore, 24 ore e 4 ore prima e
4 ore dopo l’impianto intraperitoneale di una capsula di gelatina
contenente di 10(8) unità produttrici di colonie (CFU) di S. aureus.
Sono stati raccolti dei campioni di sangue a vari periodi di tempo
dopo l’infezione per determinare: i livelli di CFU, la conta
leucocitaria e l’attivazione metabolica dei neutrofili; sono stati anche
valutati i livelli di TNF-alfa nel siero e di IL-1beta. La dose da 0.25
mg/kg di glucano non ha avuto effetti sulla riduzione del livello di
CFU; invece, le dosi di PGG-glucano da 0.5 mg/kg, 1 mg/kg, 2
mg/kg o 4 mg/kg hanno ridotto significativamente i livelli di CFU nel
sangue 48 ore dopo l’infezione. La riduzione dei livelli ematici di
CFU è risultata correlata con conte assolute dei monociti e dei
neutrofili significativamente alte e con un aumento significativo
dell’attivazione metabolica dei neutrofili, in assenza di effetti sui
livelli di TNF-alfa e IL-1beta. Negli studi aggiuntivi sono stati valutati
gli effetti sulla mortalità e sui livelli ematici di CFU in ratti trattati
con ampicillina (un antibiotico cui lo S. aurus era resistente), PGGglucano, o entrambi gli agenti. La mortalità ed i livelli ematici di CFU
sono diminuiti maggiormente nei ratti trattati con la combinazione
PGG-glucano-ampicillina rispetto sia ai ratti di controllo trattati con
soluzione salina, sia ai ratti trattati con sola ampicillina o con solo
PGG-glucano. Ne concludiamo che in vivo (1) il PGG-glucano può
incrementare l’eliminazione di uno S. aureus resistente agli
antibiotici, (2) che questa eliminazione è accompagnata da un
aumento di monociti e neutrofili e da un potenziamento dell’attività
ossidativa microbicida dei neutrofili senza alterazioni nella risposta
delle citochine proinfiammatorie, e che (3) il PGG-glucano può
incrementare l’efficacia dei trattamenti antibiotici tradizionali.
16
Passive transfer of poly-(1-6)-beta-glucotriosyl-(1-3)-betaglucopyranose glucan protection against lethal infection in
an animal model of intra-abdominal sepsis.
RL Cisneros, FC Gibson 3rd and AO Tzianabos; Channing
Laboratory, Department of Pathology, Brigham and Women's
Hospital, Boston, Massachusetts, USA. Infect Immun 1996
Jun;64(6):2201-5
Trasferimento passivo della protezione da poli-(1-6)beta-glucotriosil-(1-3)-beta-glucopiranosio
glucano
contro infezioni letali in un modello animale di sepsi
intraaddominale.
RL Cisneros, FC Gibson 3rd and AO Tzianabos; Channing
Laboratory, Department of Pathology, Brigham and Women's
Hospital, Boston, Massachusetts, USA. Infect Immun 1996
Jun;64(6):2201-5
Previous studies have established the efficacy of soluble polymers of
poly-(1-6)-beta-glucotriosyl-(1-3)-beta-glucopyranose (PGG) glucan,
a biological-response modifier, in protecting against mortality
associated with experimentally induced peritonitis in a rat model.
PGG glucan-treated animals showed increases in total leukocyte
counts and enhanced bacterial clearance from blood. To further
explore the mechanisms) by which this agent confers protection,
studies were performed to examine whether protection could be
transferred from PGG glucan-treated animals to naive recipients via
spleen cells (SC), SC lysates, or serum. Passive-transfer experiments
indicated that the responsible factor(s) was transferable by whole SC
and SC lysates, as well as by peripheral leukocytes or serum from
animals treated with PGG glucan. The transferable factor(s) was
resistant to pronase and trypsin digestion, was heat stable at 56 or
80 degrees C, and was not removed by NH4SO4 precipitation. The
protective effect of PGG glucan was abrogated by treatment with
indomethacin, a potent inhibitor of prostaglandin synthesis.
Administration of a purified prostaglandin extract from the sera of
PGG glucan-treated animals protected against mortality in the
peritonitis model. Furthermore, treatment of rats with exogenous
synthetic prostaglandin E2 also conferred protection against
mortality. These results suggest that the protective effect exhibited
by PGG glucan in the rat peritonitis model is mediated, at least in
part, by prostaglandins.
Precedenti studi hanno dimostrato l’efficacia dei polimeri solubili
di poli-(1-6)-beta-glucotriosil-(1-3)-beta-glucopiranosio (PGG)
glucano, un modificatore della risposta biologica, nel proteggere
dalla mortalità associata alla peritonite indotta sperimentalmente
in un modello murino. Gli animali trattati
con PGG glucano hanno mostrato aumenti nella conta totale dei
leucociti ed hanno aumentato la clearance batterica ematica. Per
analizzare ulteriormente i meccanismi tramite i quali questo
agente fornisce la protezione, sono stati condotti degli studi per
esaminare se la protezione potesse essere trasferita da animali
trattati con PGG-glucano verso riceventi non sensibilizzati,
tramite le cellule della milza (SC), i lisati di SC, o il siero. Gli
esperimenti di trasferimento passivo indicano che il fattore (o i
fattori) responsabili sono trasferibili tramite gli SC ed i lisati di
SC, così come tramite i leucociti periferici o il siero proveniente
da animali trattati con PGG glucano. Il fattore (o i fattori)
responsabili si sono mostrati resistenti alla digestione con pronasi
e tripsina, stabili fino a 56 o 80 gradi C e non sono stati rimossi
con precipitazione NHS404. L’effetto protettivo del PGG glucano
è stato eliminato tramite trattamento con indometacina, un
potente inibitore della sintesi della prostaglandina. La
somministrazione di un estratto purificato di prostaglandine
proveniente dal siero degli animali trattati con PGG glucano ha
protetto contro la mortalità nel modello di peritonite. Inoltre,
anche il trattamento dei topi con prostaglandina E2 sintetica
esogena ha conferito protezione contro la mortalità. Questi
risultati suggeriscono che l’effetto protettivo mostrato dal PGG
glucano nel modello murino di peritonite è mediato, almeno in
parte, dalle prostaglandine.
Synergism between poly-(1-6)-beta-d-glucopyranosyl-(1-3)
-beta-d-glucopyranose glucan and cefazolin in prophylaxis
of staphylococcal wound infection in a guinea pig model.
Sinergia tra il poli-(1-6)-beta-d-glucopiranosil-(1-3)beta-d-glucopiranosio glucano e la cefazolina nella
profilassi dell’infezione stafilococcica delle ferite nella
cavia.
Kaiser AB, Kernodle DS. Division of Infectious Diseases, Department
of Medicine, Vanderbilt University School of Medicine, Nashville,
Tennessee 37232-2605, USA. Antimicrob Agents Chemother 1998
Sep; 42(9):2449-51
To determine whether the infection-preventing capability of the
neutrophil-activating agent poly-(1-6)-beta-D-glucopyranosyl-(1-3)beta-D-glucopyranose glucan (PGG-glucan) can be enhanced with
antibiotic prophylaxis, we administered PGG-glucan and cefazolin,
alone and in combination, to guinea pigs inoculated with isolates of
staphylococci. Guinea pigs receiving both PGG-glucan and cefazolin
had 50% infective doses that were 8- to 20-fold higher than those
obtained with cefazolin alone and 100- to 200-fold higher than those
obtained with PGG-glucan alone. PGG-glucan and cefazolin are
synergistic in their ability to prevent staphylococcal wound infection.
Kaiser AB, Kernodle DS. Division of Infectious Diseases,
Department of Medicine, Vanderbilt University School of
Medicine, Nashville, Tennessee 37232-2605, USA. Antimicrob
Agents Chemother 1998 Sep; 42(9):2449-51
Al fine di determinare se la capacità di prevenire le infezioni
dell’agente
attivatore
di
neutrofili
poli-(1-6)-beta-dglucopiranosil-(1-3)-beta-d-glucopiranosio
glucano
(PGGglucano) possa o meno essere incrementata tramite profilassi
antibiotica, abbiamo somministrato PGG-glucano e cefazolina,
da soli o in combinazione, a cavie inoculate con isolati di
staffilococchi. Le cavie che hanno ricevuto sia PGG-glucano che
cefazolina hanno presentato dosi infettive al 50% da 8 a 20
volte più elevate rispetto a quelle ottenute con sola cefazolina, e
da 100 a 200 volte più elevate rispetto a quelle ottenute col solo
PGG-glucano. Il PGG-glucano e la cefazolina sono sinergici nel
prevenire l’infezione stafilococcica delle ferite.
17
Anti-infective effect of poly-beta 1-6-glucotriosyl-beta 1-3glucopyranose glucan in vivo.
Effetto anti-infettivo del poli 1-6-glucotriosil-beta 1-3glucopiranosio glucano in vivo.
AB Onderdonk, RL Cisneros, P Hinkson and G Ostroff
Department of Pathology, Channing Laboratory, Brigham and
Women's Hospital, Boston, Massachusetts. Infect Immun 1992
Apr;60(4):1642-7
AB Onderdonk, RL Cisneros, P Hinkson and G Ostroff
Department of Pathology, Channing Laboratory, Brigham and
Women's Hospital, Boston, Massachusetts. Infect Immun 1992
Apr;60(4):1642-7
Mice challenged with Escherichia coli or Staphylococcus aureus were
protected against lethal peritonitis by the intravenous administration
of 10 micrograms of poly-beta 1-6-glucotriosyl-beta 1-3glucopyranose (PGG) glucan per animal 4 to 6 h prior to bacterial
challenge. Subsequent studies with the rat model for intraabdominal sepsis indicated that intramuscular doses of 10 to 100
micrograms per animal 24 and 4 h prior to surgical implantation of
the bacterial inoculum reduced the early mortality associated with
the peritonitis phase of this experimental disease process.
Quantitative cultures of blood obtained from challenged rats showed
that significantly fewer organisms were present in the blood of PGG
glucan-treated animals than in that of untreated animals.
Quantitative studies of leukocytes of rats and mice following a single
injection of PGG glucan showed a modest transient increase in the
total leukocyte count. The possible mechanisms by which protection
occurs in the animal model system are discussed.
Topi infettati con Escherichia coli o Staphylococcus aureus sono
stati protetti contro la peritonite letale tramite la
somministrazione endovenosa di 10 microgrammi per animale di
1-6-glucotriosil-beta 1-3-glucopiranosio (PGG) glucano da 4 a 6
ore prima dell’infezione batterica. Successivi studi con modelli
animali della sepsi intra-addominale nel ratto hanno indicato che
dosi intramuscolari da 10 a 100 microgrammi per animale 24 e 4
ore prima dell’impianto dell’inoculo batterico hanno ridotto la
mortalità precoce associata con la fase peritonitica di questo
processo patologico sperimentale. Culture quantitative di sangue
ottenuto da ratti infettati hanno mostrato che erano presenti un
numero significativamente minore di microrganismi nel sangue
dei topi trattati con PGG-glucano rispetto agli animali non
trattati. Studi quantitativi dei leucociti dei ratti e dei topi dopo
una singola iniezione di PGG glucano hanno mostrato un
modesto aumento transitorio della conta totale dei leucociti. I
possibili meccanismi tramite i quali avviene la protezione nei
modelli animali sono discussi.
Diepitopic construct of functionally and epitopically
complementary
peptides
enhances
immunogenicity,
reactivity with glucosyltransferase, and protection from
dental caries.
Un costrutto diepitopico di peptidi funzionalmente ed
epitopicamente
complementari
aumenta
l’immunogenicità, la reattività con la glucosiltransferasi
e la protezione dalle carie dentali.
Taubman MA; Holmberg CJ; Smith DJ, Department of Immunology,
The Forsyth Institute, Boston, Massachusetts 02115, USA Infect
Immun 2001 Jul;69(7):4210-6 (ISSN: 0019-9567)
Coimmunization with peptide constructs from catalytic (CAT) and
glucan-binding (GLU) domains of glucosyltransferase (GTF) of
mutans streptococci has resulted in enhanced levels of antibody to
the CAT construct and to GTF. We designed and synthesized a
diepitopic construct (CAT-GLU) containing two copies of both CAT (B
epitope only) and GLU (B and T epitope) peptides. The
immunogenicity of this diepitopic construct was compared with that
of individual CAT and GLU constructs by immunizing groups of
Sprague-Dawley rats subcutaneously in the salivary gland vicinity
with the CAT-GLU, CAT, or GLU construct or by treating rats by
sham immunization. Levels of serum immunoglobulin G (IgG)
antibody to GTF or CAT in the CAT-GLU group were significantly
greater than in GLU- or CAT-immunized groups. Immunization with
CAT-GLU was compared to coimmunization with a mixture of CAT
and GLU in a second rodent experiment under a similar protocol.
CAT-GLU immunization resulted in serum IgG and salivary IgA
responses to GTF and CAT which were greater than after
coimmunization. Immunization with the diepitopic construct and
communization with CAT and GLU constructs showed proliferation of
T lymphocytes to GTF. Immunization with either the CAT or GLU
construct has been shown to elicit significant protection in a rodent
dental caries model. Similarly in this study, the enhanced response
to GTF after immunization with the CAT-GLU construct resulted in
protective effects on dental caries. Therefore, the CAT-GLU
diepitopic construct can be a potentially important antigen for a
caries vaccine, giving rise to greater immune response than after
immunization with CAT, GLU, or a mixture of the two.
AB Onderdonk, RL Cisneros, P Hinkson and G Ostroff
Department of Pathology, Channing Laboratory, Brigham and
Women's Hospital, Boston, Massachusetts. Infect Immun 1992
Apr;60(4):1642-7
La coimmunizzazione con costrutti peptidici composti dai domini
catalitico (CAT) e di legame del glucano (GLU) della
glucosiltransferasi (GTF) di streptococchi mutanti, ha generato
un incremento del livello di antricorpi anti-costrutti CAT e antiGTF. Noi abbiamo progettato e sintetizzato un costrutto
diepitopico (CAT-GLU) contenente due copie ciascuno dei
peptidi CAT (solo l’epitopo B) e GLU (epitopi B e T).
L’immunogenicità di questo costrutto diepitopico è stata
confrontata con quella dei costrutti singoli di CAT e GLU tramite
l’immunizzazione sottocutanea in prossimità della ghiandola
salivare di gruppi di ratti di Sprague-Dawley con i costrutti CATGLU, CAT oppure GLU, ovvero trattando i ratti con falsa
immunizzazione. I livelli nel siero di immunoglobulina G (IgG)
anticorpale verso GTF o CAT erano significativamente maggiori
nei gruppi CAT-GLU rispetto ai gruppi immunizzati GLU o CAT.
L’immunizzazione con CAT-GLU è stata confrontata con la
coimmunizzazione con una miscela di CAT e GLU in un secondo
esperimento con roditori utilizzando un protocollo simile.
L’immunizzazione CAT-GLU ha prodotto risposte al GTF ed al
CAT, in termini di IgG del siero e IgA salivare, che sono risultate
maggiori rispetto a quelle conseguenti alla coimmunizzazione.
L’immunizzazione
col
costrutto
diepitopico
e
la
coimmunizzazione con i costrutti CAT e GLU ha mostrato
proliferazione dei linfociti T anti-GTF. L’immunizzazione con
costrutti CAT o GLU si è dimostrata efficace nel fornire una
protezione significativa in un modello di carie dentale nei
roditori. Analogamente, in questo studio l’incrementata risposta
al GTF successiva all’immunizzazione con il costrutto CAT-GLU
ha avuto effetti protettivi sulle carie dentali. Pertanto, il
costrutto diepitopico CAT-GLU può essere un antigene
potenzialmente importante per un vaccino contro la carie,
poiché da una maggior risposta immunitaria rispetto
all’immunizzazione con CAT, GLU o una miscela dei due.
18
Prophylaxis with the immunomodulator PGG glucan
enhances antibiotic efficacy in rats infected with antibioticresistant bacteria.
La profilassi con l’immunomodulatore PGG glucano
incrementa l’efficacia degli antibiotici in ratti infettati
con batteri antibiotico-resistenti.
A. O. Tzianabos and R. L. Cisneros; Channing Laboratory Brigham
and Women's Hospital, Harvard Medical School Boston,
Massachusetts 02115, USA. Ann N Y Acad Sci. 1996 Oct 25;
797:285-7.
A. O. Tzianabos and R. L. Cisneros; Channing Laboratory
Brigham and Women's Hospital, Harvard Medical School Boston,
Massachusetts 02115, USA. Ann N Y Acad Sci. 1996 Oct 25;
797:285-7.
The emergence of multiple antibiotic-resistant microorganisms has
led to a search for alternatives to traditional therapeutic regimens.
PGG glucan is a soluble beta-glucan immunomodulator that
selectively enhances the microbicidal activities of neutrophils and
macrophages without stimulating proinflammatory cytokine
production. In the present studies, we examined the ability of PGG
glucan to act in concert with antibiotics to decrease mortality in a rat
model of intraabdominal sepsis using antibiotic-resistant bacteria as
infectious inocula. Results of these studies demonstrated that
prophylaxis with PGG glucan in combination with antibiotics provided
enhanced protection against lethal challenge with Esherichia coli or
Staphylococcus aureus as compared with the use of antibiotics
alone.
L’emergere di molti microorganismi antibiotico-resistenti ha
portato a ricercare delle alternative ai regimi terapeutici
tradizionali.
Il
PGG
glucano
è
un
beta-glucano
immunomodulatore solubile che aumenta selettivamente le
attività microbicide dei neutrofili e dei macrofagi senza stimolare
la produzione di citochine proinfiammatorie. Nel presente studio
abbiamo esaminato la capacità del PGG glucano di agire di
concerto con gli antibiotici nel decrescere la mortalità in un
modello animale di sepsi intraaddominale nel ratto ottenuto
utilizzando batteri antibiotico-resistenti come infettanti. I risultati
di questi studi hanno dimostrato che la profilassi con PGG
glucano
in
associazione
con
antibiotici
ha
fornito
un’incrementata protezione contro infezioni letali da E. coli o da
S. aureus, rispetto all’uso del solo antibiotico.
Protective effect of beta-glucan against
Streptococcus pneumoniae infection in mice.
Effetto protettivo del beta-glucano contro l’infezione
sistemica da Streptococcus pneumoniae nei topi.
systemic
Hetland G; Ohno N; Aaberge IS; Lovik M Department of
Environmental Medicine, National Institute of Public Health, Oslo,
Norway. FEMS Immunol Med Microbiol 2000 Feb;27(2):111-6 (ISSN:
0928-8244)
Hetland G; Ohno N; Aaberge IS; Lovik M Department of
Environmental Medicine, National Institute of Public Health,
Oslo, Norway. FEMS Immunol Med Microbiol 2000
Feb;27(2):111-6 (ISSN: 0928-8244)
The antimicrobial effect of soluble beta-1,3-D-glucan from
Sclerotinia sclerotiorum (SSG) was examined in mice experimentally
infected intraperitoneally (i.p.) with Streptococcus pneumoniae
serotypes 4 and 6B. SSG was administered i.p. either 3 days before
challenge or 3-48 h after challenge. The number of bacteria in blood
samples and the mouse survival rates were recorded. Pre-challenge
SSG administration protected dose-dependently against both S.
pneumoniae type 4 and 6B infections. SSG injected 24 h postchallenge had a curative effect against type 6B but not type 4
pneumococcal infection. The data demonstrate that SSG
administered systemically protects against pneumococcal infection in
mice.
L’effetto antimicrobico del beta-1,3-D-glucano estratto da
Sclerotinia sclerotiorum (SSG) è stato esminato in topi infettati
sperimentalmente per via intraperitoneale (i.p.) con i sierotipi 4
e 6B di Streptococcus pneumoniae. SSG è stato somministrato
i.p. o 3 giorni prima dell’infezione o 3-48 ore dopo l’infezione.
Sono stati registrati il numero di batteri presenti nei campioni di
sangue e i tassi di sopravvivenza dei topi. La somministrazione
di SSG pre-infezione ha protetto in modo dose-dipendente sia
dall’infezione col tipo 4 di S. pneumoniae, sia dall’infezione col
tipo 6B. SSG iniettato 24 ore dopo l’infezione ha avuto un
effetto terapeutico verso il tipo 6B ma non verso il tipo 4 di
infezione pneumococcica. I dati dimostrano che SSG
somministrato per via sistemica è protettivo contro le infezioni
pneumococciche nei topi.
The protective effect of beta 1-3D-glucan-derivatized
plastic beads against Escherichia coli infection in mice.
L’effetto protettivo di granuli plastici derivatizzati con
beta 1-3D-glucano verso l’infezione da Escherichia coli
nel topo.
Seljelid R; Rasmussen LT; Larm O; Hoffman J, Scand J Immunol
1987 Jan;25(1):55-60 (ISSN: 0300-9475)
Pretreatment with beta-1,3-D-glucan-derivatized plastic beads
conferred strong protection against Escherichia coli infection in
mice. The protective effect showed a dose-response relationship to
the amount of beads injected and was dependent on the time point
of the injection relative to the infection with E. coli. A similar
protection could be obtained in nude mice. Experiments with
radioactively labelled bacteria as well as beads indicated a systemic
effect of the beads. Macrophages extracted from animals treated
with glucan plastic beads appeared highly stimulated. This was also
true of cells that did not contain beads and presumably therefore
not glucan, which seems to indicate a soluble stimulatory factor
Seljelid R; Rasmussen LT; Larm O; Hoffmann J, Scand J
Immunol 1987 Jan;25(1):55-60 (ISSN:0300-9475)
Il pretrattamento con granuli plastici derivatizzati con beta 1-3D-glucano ha conferito una forte protezione contro l’infezione da
Escherichia coli nei topi. L’effetto protettivo ha mostrato una
relazione dose-risposta alla quantità di granuli iniettati ed una
dipendenza dall’intervallo di tempo intercorrente tra l’iniezione e
l’infezione con E. coli. Una protezione simile può essere ottenuta
nei topi nudi. Gli esperimenti con batteri e granuli marcati
radioattivamente hanno mostrato un effetto sistemico dei
granuli. I macrofagi estratti dagli animali trattati con i granuli
plastici al glucano apparivano altamente stimolati. Questo
valeva anche per le cellule non contenenti granuli, e pertanto
presumibilmente nemmeno glucano, il che sembra indicare la
presenza di un fattore stimolatorio solubile.
19
GLUCAN
Re-epithalializator and healer of tissue
activities
HO
O
CH 2 OCH 2 COO
-
Na
+
O
OH
HO
O
CH 2 OH
OH
20
O
Promotion of Wound repair in Mice by Application of Glucan.
S.J Leibovich and D. Danon, The section of biological ultrastructure,
the weizmann institute of science, P.O.B. 26, Rehovot, Israel Journal
of the Reticuloendothelial Society, Vol.27, No1,January 1980
La promozione della guarigione della ferita nei topi
attraverso l’applicazione di glucano.
S.J Leibovich and D. Danon, The section of biological ultrastructure,
the weizmann institute of science, P.O.B. 26, Rehovot, Israel Journal
of the Reticuloendothelial Society, Vol.27, No1,January 1980
The effects of topical application of a variety of substances (glucan,
carrageenan, levan, inulin, dextran, starch and talcum powder) on
the repair of skin wounds in SWR mice were examined. Of the
substance tested, only glucan showed any marked beneficial effects.
Glucan –treated wounds showed a higher number of macrophages
in the early, inflammatory stage of repair, with fever
polymorphonuclear neutrophilic leukocytes then did control wounds.
Both re-epithalialization and the onset of fibroplasia commenced at
an early stage in glucan treated wounds then in control wounds.
Five days following wounding glucan treated wounds were generally
completely rehepitelializeted, while control wounds were not the
organization of fibroblats in glucan treaded wounds was more
advanced at 5 and 7 days following injury, and the extent of
fibroplasias was also greater. By 10 days following injury glucan
treated wounds were completely re-epithelializated , as were control
wounds treated with medium alone inulin dextran, starch or talc.
Carrageenan and levan-treated wounds showed impaired reepitheliliazation.
Only
occasional
residual
glucan-containg
macrophages were present in the glucan-treated wounds at this
stage. In glucan treated wounds, no formation of granulomas was
observed up to one month following wounding.
Sono stati esaminati gli effetti di applicazioni topiche di una varietà
di sostanze (glucano, carragenina, levan, inulina, destrano, amido e
polvero di talco) sulla guarigione di ferite della pelle di topi SWR.
Tra le sostanze testate, solamente il glucano ha mostrato degli
effetti benefici significativi. Le ferite trattate con glucano hanno
mostrato un più alto numero di macrofagi nella prima fase
infiammatoria della guarigione, con più leucociti polimorfonucleati
neutrofili di quanto abbiano mostrato le ferite di controllo. Sia la
riepitelizzazione sia lo sviluppo di tessuto fibroso sono comparsi
prima nelle ferite trattate con glucano che in quelle di controllo. Le
ferite testate con glucano erano generalmente completamente
riepitelizzate dopo cinque giorni dalla lesione, mentre le ferite di
controllo non lo erano e l’organizzazione dei fibroblasti nelle ferite
trattate con glucano era più avanzata a 5 e 7 giorni dopo la lesione,
e anche l’organizzazione dei fibroblasti e la quantità di fibroplasia
era maggiore. Dopo 10 giorni dalla lesione, le ferite trattate con
glucano erano completamente riepitelizzate, così come le ferite di
controllo trattate solamente con inulina, destrano, amido o talco. Le
ferite trattate con carragenina o levan hanno mostrato una
riepitelizzazione ridotta. Solo occasionali macrofagi residui
contenenti glucani erano presenti nelle ferite trattate con glucano in
questa fase. Nelle ferite trattate con glucano nessuna formazione di
granulomi è stata osservata fino ad un mese dalla lesione.
Effect of macrophage stimulation on collagen biosynthesis
in the healing wound.
Effetto della stimolazione macrofagica sulla biosintesi del
collagene durante la guarigione della ferita.
Immunomodulators that enhance macrophage function have been
shown to be beneficial in a number of wound-healing models in
humans and in experimental animals. The exact mechanism of this
improved healing is unclear. To assess the role of collagen
biosynthesis, the immunomodulator glucan phosphate was utilized
in two murine models of wound healing, i.e., colon anastomosis and
full-thickness skin incision. Tensile strength was evaluated using
computer-assisted constant velocity tensiometry. Collagen
biosynthesis was determined by assaying hydroxyproline content of
wound
hydrolysates
by
N-(9-fluorenyl)methoxycarbonyl/ophthalaldehyde
high-performance
liquid
chromatography.
Experimental animals were treated with (1-3)-beta-D-glucan
phosphate (250 mg/kg) intravenously 24 hours prior to colon
anastomosis or skin incision. A second dose of glucan phosphate
was given immediately postoperatively. Control animals received
dextrose and water (5% w/v) intravenously. Tensile strength and
hydroxyproline content were measured on postoperative Day 3. In
the skin wound model, glucan phosphate treatment increased (P <
0.05) tensile strength by 42 per cent (342.5 +/- 12.2 vs 241.8 +/4.8 g), and hydroxyproline content was increased by 23.5 per cent
(242.0 +/- 14.4 vs 196.8 +/- 10.5 pmol/microg; P < 0.05). In the
glucan phosphate group, colon tensile strength was significantly (P
< 0.05) increased by 34 per cent (34.2 +/- 2.3 g vs 45.8 +/- 2.1 g),
and hydroxyproline content was increased by 7 per cent (47.45 +/3.31 vs 44.34 +/- 3.74 pmol/microg). These data indicate that
macrophage modulation with glucan phosphate will increase tensile
strength in experimental colon and skin wounds. In addition, we
observed a positive correlation between glucan phosphate
treatment, wound tensile strength, and collagen biosynthesis.
Gli immunomodulatori che potenziano la funzione macrofagica
hanno mostrato di essere benefici in numerosi modelli di
guarigione della ferita nell’uomo e nell’animale da esperimento.
L’esatto meccanismo di questo miglioramento della guarigione
non è chiaro. Per verificare il ruolo della biosintesi del collagene,
è stato utilizzato l’immunomodulatore glucanfosfato in due
modelli murini di guarigione della ferita, ossia l’anastomosi del
colon e l’incisione della pelle a tutto spessore. Lo resistenza di
trazione è stata valutata usando la tensiometria computerizzata a
velocità costante. La biosintesi del collagene è stata determinata
misurando il contenuto di idrossiprolina degli idrolisati di ferite
attraverso cromatografia liquida ad alte prestazioni di N-(9fluorenil) metocsicarbonil/oftalaldeide. Gli animali da esperimento
sono stati trattati con iniezione endovenosa di glucano (1-3)beta-D fosfato (250 mg/kg) 24 ore prima dell’anastomosi del
colon o dell’incisione della pelle. Una seconda dose di
glucanfosfato è stata somministrata immediatamente dopo
l’operazione. Gli animali di controllo hanno ricevuto destrosio e
acqua (5% w/v) per via endovenosa. La forza di trazione e il
contenuto di idrossiprolina sono stati misurati il terzo giorno
postoperatorio. Nel modello della ferita della pelle, il trattamento
di glucanfosfato ha aumentato (P < 0.05) la resistenza a trazione
del 42% (342.5 +/- 12.2 vs 241.8 +/- 4.8 g), e il contenuto di
idrossiprolina del 23.5% (242.0 +/- 14.4 vs 196.8 +/- 10.5
pmol/microg; P < 0.05). Nel gruppo trattato con glucanfosfato,
la resistenza a trazione del colon è aumentata significativamente
(P < 0.05) del 34% (34.2 +/- 2.3 g vs 45.8 +/- 2.1 g), e il
contenuto di idrossiprolina del 7 % (47.45 +/- 3.31 vs 44.34 +/3.74 pmol/microg). Questi dati indicano che la modulazione
macrofagica con glucanfosfato è in grado di aumentare la
resistenza a trazione nelle ferite sperimentali del colon e della
pelle. Inoltre, è stata riscontrata una correlazione positiva tra il
trattamento con glucanfosfato, la resistenza a trazione della
ferita e la biosintesi del collagene.
Portera CA; Love EJ; Memore L; Zhang L; Muller A; Browder W;
Williams DL; Department of Surgery, James H. Quillen College of
Medicine, East Tennessee State University, Johnson City 376140575, USA. Am Surg 1997 Feb;63(2):125-31 (ISSN: 0003-1348)
Portera CA; Love EJ; Memore L; Zhang L; Muller A; Browder W;
Williams DL; Department of Surgery, James H. Quillen College of
Medicine, East Tennessee State University, Johnson City 376140575, USA. Am Surg 1997 Feb;63(2):125-31 (ISSN: 0003-1348)
21
Effect of enhanced macrophage function on early wound
healing.
Effetto del potenziamento della funzione macrofagica
nella prima fase di guarigione della ferita.
Browder W; Williams D; Lucore P; Pretus H; Jones E; McNamee R
Department of Surgery, Tulane University School of Medicine, New
Orleans, LA 70112. Surgery 1988 Aug;104(2):224-30 (ISSN:
0039-6060)
Browder W; Williams D; Lucore P; Pretus H; Jones E; McNamee
R Department of Surgery, Tulane University School of Medicine,
New Orleans, LA 70112. Surgery 1988 Aug;104(2):22430 (ISSN: 0039-6060)
Although the macrophage is important to wound healing, research
has focused on its relationship to fibroblast and collagen synthesis.
This study was designed to assess effects of enhanced macrophage
function on early wound healing, before established collagen
synthesis. Sprague-Dawley rats had dorsal incisions after one of
three treatment regimens: (1) saline solution, 0.5 ml administered
intravenously, (2) intravenous glucan, a macrophage stimulant, 20
mg; (3) topical glucan, 20 mg. Intravenous therapy was
administered 24 hours before and after incision. Breaking strength
was significantly increased (p less than 0.01) by both intravenous
glucan (49.8 +/- 5.5 gm) and topical glucan (59.7 +/- 5.6 gm) on
the fourth day after incision, compared with controls (22.0 +/- 2.6
gm). Similar results occurred on the seventh day after incision.
Although formalin fixation significantly enhanced breaking strength
in fresh control wounds (22.0 +/- 2.6 vs 39.5 +/- 2.2 gm), no
increase occurred in wounds treated with intravenous glucan (49.8
+/- 5.0 vs 55.3 +/- 6.4 gm), indicating maximal cross-linking of
collagen. Collagen synthesis, reflected by tritiated proline uptake,
was no different in control versus glucan groups. Supernatants from
control
or
glucan-activated
macrophages
were
injected
intraperitoneally or applied topically in the rat model. Activated
supernatant, both intraperitoneal and topical, resulted in increased
breaking strength on the fourth day after incision. Formalin fixation
did not increase breaking strength in the activated supernatant
groups. We conclude that enhanced macrophage function increases
early wound breaking strength. This effect appears unrelated to
collagen synthesis but may be related to increased cross-linking of
collagen. Similar effects are seen with activated macrophage
secretory products administered intraperitoneally or topically.
Sebbene i macrofagi siano ritenuti importanti nella del collagene
ma potrebbe essere collegato all’aumento di guarigione di una
ferita, la ricerca si è focalizzata sui nessi dei macrofagi stessi con
i fibroblasti e con la sintesi del collagene. Questo studio è stato
pensato per quantificare gli effetti del potenziamento della
funzione macrofagica nella prima fase della guarigione della
ferita, prima che si stabilisca la sintesi del collagene. Dei ratti
Sprague-Dowley hanno subito una incisione dorsale dopo uno
dei seguenti tre trattamenti: (1) fisiologica, 0.5 ml somministrati
per via endovenosa, (2) glucano endovenoso, uno stimolatore di
macrofagi, 20 mg, (3) glucano topico, 20 mg. La terapia
endovenosa è stata somministrata 24 ore prima e dopo
l’incisione. La resistenza a rottura è aumentata in maniera
significativa (p < 0.01) sia per il glucano endovenoso (49.5 +/5.5 gm) sia per il glucano topico (59.7 +/- 5.6 gm) al quarto
giorno dopo l’incisione, se confrontato con il gruppo di controllo
(22.0 +/- 2.6 gm). Risultati simili sono stati osservati al settimo
giorno dopo l’incisione. Sebbene il fissaggio in formalina abbia
aumentato significativamente la resistenza a rottura delle ferite
fresche nel gruppo di controllo (22.0 +/- 2.6 gm vs 39.5 +/-2.2
gm), non è stato riscontrato un aumento nelle ferite trattate con
glucano endovenoso (49.8 +/- 5.0 vs 55.3 +/- 6.4 gm), fatto che
indica un massimo cross-linking del collagene. La sintesi del
collagene, evidente dall’assorbimento di prolina marcata con
trizio radioattivo, non era differente tra il gruppo di controllo e
quelli trattati con glucano. I surnatanti dai macrofagi di controllo
o dai macrofagi attivati dal glucano sono stati iniettati per via
intraperitoneale o applicati per via topica nel modello di ratto. I
supernatanti attivati, sia per via peritoneale che per via topica,
hanno aumentato la resistenza a rottura al quarto giorno dopo
l’incisione. La fissazione in formalina non ha aumentato la
resistenza a rottura nel gruppo di supernatanti attivati.
Concludiamo che il potenziamento della funzione macrofagica
aumenta la resitenza a rottura nella prima fase di guarigione
della ferita. Questo effetto appare non correlato alla sintesi
cross-linking del collagene. Effetti simili sono stati osservati con
prodotti di secrezione dei macrofagi attivati somministrati per via
peritoneale o topica.
22
Normal human fibroblasts express pattern recognition
receptors for fungal (1-->3)-beta-D-glucans.
Kougias P; Wei D; Rice PJ; Ensley HE; Kalbfleisch J; Williams DL;
Browder. IW Departments of Surgery, James H. Quillen College of
Medicine, Johnson City, Tennessee 37614, USA. Infect Immun 2001
Jun;69(6):3933-8 (ISSN: 0019-9567)
Fungal cell wall glucans nonspecifically stimulate various aspects of
innate immunity. Glucans are thought to mediate their effects via
interaction with membrane receptors on macrophages, neutrophils,
and NK cells. There have been no reports of glucan receptors on
nonimmune cells. We investigated the binding of a water-soluble
glucan in primary cultures of normal human dermal fibroblasts
(NHDF). Membranes from NHDF exhibited saturable binding with an
apparent dissociation constant (K(D)) of 8.9 +/- 1.9 microg of
protein per ml and a maximum binding of 100 +/- 8 resonance
units. Competition studies demonstrated the presence of at least
two glucan binding sites on NHDF. Glucan phosphate competed for
all binding sites, with a K(D) of 5.6 microM (95% confidence interval
[CI], 3.0 to 11 microM), while laminarin competed for 69% +/- 6%
of binding sites, with a K(D) of 3.7 microM (95% CI, 1.9 to 7.3
microM). Glucan (1 microg/ml) stimulated fibroblast NF-kappaB
nuclear binding activity and interleukin 6 (IL-6) gene expression in a
time-dependent manner. NF-kappaB was activated at 4, 8, and 12
h, while IL-6 mRNA levels were increased by 48% at 8 h. This is the
first report of pattern recognition receptors for glucan on human
fibroblasts and the first demonstration of glucan binding sites on
cells other than leukocytes. It also provides the first evidence that
glucans can directly modulate the functional activity of NHDF. These
results provide new insights into the mechanisms by which the host
recognizes and responds to fungal (1-->3)-beta-D-glucans and
suggests that the response to glucans may not be confined to cells
of the immune system.
I fibroblasti umani normali esprimono recettori di “pattern
recognition” per i gli (1-->3)-beta-D-glucani fungini.
Kougias P; Wei D; Rice PJ; Ensley HE; Kalbfleisch J; Williams DL;
Browder. IW Departments of Surgery, James H. Quillen College
of Medicine, Johnson City, Tennessee 37614, USA. Infect Immun
2001 Jun;69(6):3933-8 (ISSN: 0019-9567)
I glucani della parete cellulare dei funghi stimolano in maniera
aspecifica vari aspetti dell’immunità innata. Si ritiene che i
glucani agiscano tramite l’interazione con i recettori di
membrana sui macrofagi, sui neutrofili e sulle cellule NK. Non è
stata mai riportata la presenza di recettori per i glucani su
cellule non immunitarie. Noi abbiamo investigato il legame di un
glucano idrosolubile usando culture primarie di fibroblasti
normali del derma umano (NHDF). Le membrane degli NHDF
hanno mostrato un legame saturabile con una costante di
dissociazione apparente (K(D)) di 8.9 +/- 1.9 microgrammmi di
proteina per ml ed un legame massimo di 100 +/- 8 unità di
risonanza. Studi comparativi hanno mostrato la presenza di
almeno due siti di legame per il glucano sugli NHDF. Il glucan
fosfato competeva per tutti i siti di legame, con una K(D) di 5.6
microM (intervallo di confidenza [CI] al 95%, da 3 ad 11
microM), mentre la laminarina competeva per il 69% +/- 6%
dei siti di legame, con una K(D) di 3.7 microM (CI 95%, da 1.9 a
7.3 microM). Il glucano (1 microg/ml) ha stimolato in maniera
tempo-dipendente l’attività di legame nucleare NF-kappaB e
l’espressione del gene per l’ intereleuchina-6 (IL-6) dei
fibroblasti. NF-kappaB è stata attivata a 4, 8 e 12 ore, mentre i
livelli di mRNA per IL-6 sono cresciuti del 48% ad 8 ore. Questo
è il primo rapporto riguardo ai recettori di “pattern recognition”
per il glucano sui fibroblasti umani e la prima dimostrazione
della presenza di siti di legame per il glucano su cellule diverse
dai leucociti. Inoltre, questo studio evidenzia per la prima volta
che i glucani possono modulare direttamente l’attività funzionale
degli NHDF. Questi risultati forniscono nuovi approfondimenti
riguardo ai meccanismi tramite i quali l’organismo riconosce e
risponde agli (1-->3)-beta-D-glucani fungini e suggeriscono che
la risposta ai glucani può non essere limitata alle cellule del
sistema immunitario.
23
The beneficial effect of enhanced macrophage function on
the healing of bowel anastomoses.
Compton R; Williams D; Browder W , Department of Surgery, East
Tennessee State University, Johnson City 37614, USA. Am Surg
1996 Jan;62(1):14-8 (ISSN: 0003-1348)
Effetto benefico del potenziamento della funzione
macrofagica
nella
guarigione
dell’anastomosi
dell’intestino.
Compton R; Williams D; Browder W , Department of Surgery,
East Tennessee State University, Johnson City 37614, USA. Am
Surg 1996 Jan;62(1):14-8 (ISSN: 0003-1348)
Inadequate healing and subsequent leakage of bowel anastomoses
are serious postoperative complications in abdominal surgery.
Previous studies have demonstrated the macrophage to be a key
cell in the physiology of wound healing. The current study was
undertaken to evaluate the effects of enhanced macrophage
function on the healing of bowel anastomoses. Sprague-Dawley rats
(250 gm) underwent laparotomy and jejunojejunostomy following IV
treatment with glucan (100 mg per kg), a potent macrophage
stimulant, or 5 per cent dextrose 24 hours before surgery and again
on the day of surgery. Animals were killed and the anastomoses
underwent wound tensiometry on Day 3 using a computer-assisted
constant velocity tensiometer. The glucan treated animals had a
significantly greater anastomotic breaking strength (88.5 gm +/10.7 versus 45.45 gm +/- 5.1) (P < 0.01). Formalin fixation
increased the breaking strength of the untreated anastomosis but
not of the treated anastomosis (92.9 gm +/- 11.77 versus 92.3 +/12.44). Analysis of macrophage supernatant for the growth factors
epidermal growth factor (EGF), platelet derived growth factor
(PDGF), and transforming growth factor-beta (TGF-beta) was
accomplished by immunoblot assay. Results indicated no difference
in the presence of EGF in the stimulated and unstimulated
macrophage supernatants. PDGF and TGF-beta were decreased in
the stimulated macrophage supernatants. We conclude that 1)
Enhanced macrophage function had a beneficial effect on the early
tensile strength of bowel anastomoses. 2) Effects of the activated
macrophage on bowel anastomoses may not be related to secretion
of conventional growth factors. 3) Immunopharmacologic agents
that enhance macrophage function may be an important adjunct to
surgical therapy requiring bowel anastomosis.
Guarigioni inadeguate e conseguenti perdite delle anastomosi
dell’intestino sono serie complicazioni postoperatorie nella
chirurgia addominale. Ricerche passate hanno dimostrato che i
macrofagi sono le cellule-chiave nella fisiologia della guarigione
delle ferite. Il presente studio è stato intrapreso per valutare gli
effetti del potenziamento della funzione macrofagica sulla
guarigione delle anastomosi dell’intestino. Dei ratti SpragueDowley (250 gm) sono stati sottoposti a laparotomia e
digiunostomia dopo il trattamento IV con glucano (100 mg per
kg), un potente stimolante microfagico, o con 5% di destrosio
24 ore prima e nel giorno stesso dell’operazione chirurgica. Gli
animali sono stati sacrificati e le anastomosi sono state testate a
trazione con un tensiometro computerizzato a velocità costante.
Gli animali trattati con glucano avevano una resistenza a rottura
della ferita significativamente più grande (88.5 gm +/- 10.7 vs
45.45 gm +/- 5.1) (P < 0.01). La fissazione con formalina ha
aumentato la forza di rottura delle anastomosi non trattate ma
non quella delle anastomosi trattate (92.9 gm +/- 11.77 vs 92.3
gm +/- 12.44). L’analisi dei surnatanti macrofagici per
individuare i fattori di crescita: fattore di crescita epidermico
(EFG), fattore di crescita derivato delle piastrine (PDGF) e
fattore di crescita beta (TGF-beta) è stata condotta con
dosaggio immunoblot*. I risultati non hanno indicato differenze
nella presenza dell’EGF sia nei surnatanti di macrofagi stimolati
sia in quelli non stimolati. Il PDGF ed il TGF-beta sono diminuiti
nei surnatanti di macrofagi stimolati. Noi concludiamo che 1) il
potenziamento della funzione macrofagica ha avuto un effetto
benefico sulla resistenza a trazione dell’anastomosi dell’intestino
nella prima fase di guarigione, 2) gli effetti dei macrofagi attivati
sull’anastomosi dell’intestino possono essere non correlati alla
secrezione di fattori di crescita convenzionali, 3) gli agenti
immunofarmacologici che potenziano la funzione macrofagica
possono essere un importante integrazione alla terapia
chirurgica richiesta nell’anastomosi dell’intestino.
* Trasferimento elettroforetico di proteine separate su un gel di
poliacrilammide dal gel a una membrana di nitrocellulosa, su cui
vengono immobilizzate. La presenza di una specifica proteina
viene quindi evidenziata mediante reazione con il relativo
anticorpo opportunamente marcato.
24
ANNEX A
LIST OF SOME STUDIES ON BETA GLUCAN
ABDOMINAL OR THORACIC SURGERY - HARVARD MEDICAL SCHOOL (USA) "There
were no adverse drug experiences....is safe and appears to be effective in the further
reduction of the morbidity and cost of major surgery."
BACTERIAL INFECTIONS - BAYLOR COLLEGE OF MEDICINE; Wyde, P., "Beta-1,3glucan activity in mice: intraperitoneal and oral applications." " Beta glucan, through the
stimulation of host defense systems, creates a more supportive environment within the body
to assist the primary killing action of the conventional agent."
CANCER - CHEM PHARM BULL (Japan); "Antitumor and immunomodulating activities of a
beta-glucan...."
CANCER, LUNG AND BREAST - NATIONAL CANCER INST (USA); "The initial 9 patients
studied had malignant melanoma, adenosquamous carcinoma of the lung, or carcinoma of
the breast. Control and experimental lesions were injected: subsequently biopsies were
performed at varying intervals for histologic evaluation. Always when glucan or glucan and
RF fraction were administered intralesionally, the size of the lesion was strikingly reduced in
as short a period as 5 days. This reduction was associated with necrosis of the tumor and a
monocytic infiltrate. In small lesions, resolution was complete, whereas in large lesions,
resolution was partial.
CANCER - MAYO CLINIC (USA); "..... beta-glucan interacts with vitronectin and stimulates
tumor necrosis factor alpha release from macrophage's."
CANCER - UNIVERSITY OF TROMSO (Norway); "Macrophages stimulated by an insoluble
beta 1-3-D-glucan from yeast cell walls were able to destroy tumour cells as measured by
the release of radioactive label from prelabelled 14C-thymidine cells. Target cells were B-16
melanoma, P-815 mastocytoma, and the L-929 cell line. A significant target cell killing by
macrophages stimulated by glucan was observed after 72-96 h."
CANDIDA ALBICANS - DEPARTMENT OF SURGERY, TULANE UNIVERSITY; "Protection
against C. albicans was observed in the glucan-treated groups. ...These observations
suggest that Biologic Response Modifiers such as glucan may be effectively employed in
patients who are at risk for post-operative infections."
CHOLESTEROL (LDL) - OTTAWA CIVIC HOSPITAL (Canada); "CONCLUSIONS: The
main component of the soluble fiber of oats, beta-glucan, significantly reduced the total and
LDL cholesterol levels of hypercholesterolemic adults without changing HDL cholesterol."
CHOLESTEROL - DEPARTMENT OF AGRICULTURE (USA); "Beneficial reduction of
cholesterol was obtained with modest amounts....."
DIABETES - OTTAWA CIVIC HOSPITAL (Canada); "A diet rich in beta-glucan may
therefore be of benefit in the regulation of plasma glucose levels in subjects with Type 2
diabetes."
DIABETES - NESTLAE RESEARCH CENTER (Switzerland); "Diabetic individuals can
benefit from diets that are high in beta-glucan, .."
E. COLI - TULANE UNIVERSITY (USA, La); "Glucan therapy also increased bone marrow
proliferation. We conclude that (1) glucan enhances peritoneal neutrophil levels, (2)
Peripheral blood neutrophils are increased following glucan and E. coli, (3) ip glucan
increase bone marrow proliferation, .... Thus, the beneficial effect of glucan is mediated not
only by activated macrophages, but also by the neutrophilic leukocyte."
E. COLI, STAPHYLOCOCCUS - DEPARTMENT OF PATHOLOGY, BRIGHAM AND
WOMEN'S HOSPITAL (USA, Mass) "Mice challenged with Escherichia coli or
Staphylococcus aureus were protected against lethal peritonitis by the intravenous
administration of 10 micrograms of poly-beta 1-6-glucotriosyl-beta 1-3-glucopyranose (PGG)
glucan per animal 4 to 6 h prior to bacterial challenge."
FUNGAL INFECTION - TULANE UNIVERSITY; "The broad spectrum of
immunopharmacological activities of glucan includes not only the modification of certain
bacterial, fungal, viral and parasitic infections, but also inhibition of tumor growth."
HEPATITIS, VIRAL - SCIENCE (1980);"Thus glucan is capable of increasing survival,
inhibiting hepatic necrosis, and maintaining an activated state of phagocytic activity in mice
challenged with [mouse hepatitis virus strain] MHV-A59."
HERPES SIMPLEX 1 - PLANTA MED., 62:4, 301-7. (1996); "The antiviral effect of
scleroglucan seems to be related to its binding with membrane glycoproteins of HSV-1
particles which impedes the complex interactions of the virus with the cell plasma
membrane."
HIGH RISK SURGICAL PATIENTS - HARVARD MEDICAL SCHOOL (USA); "Patients who
received PGG-glucan had significantly fewer infectious complications (3.4 infections per
infected patient vs. 1.4 infections per infected patient, p = 0.05), decreased intravenous
antibiotic requirement (10.3 days vs. 0.4 days, p = 0.04) and shorter intensive care unit
length of stay (3.3 days vs. 0.1 days, p = 0.03). CONCLUSIONS: PGG-glucan is safe and
appears to be effective in the further reduction of the morbidity and cost of major surgery.
INFECTION PREVENTION - GYNECOLOGY & OBSTETRICS, 177:383-388. (1993); "The
incidence of hospital pneumonia of 55% and sepsis of 35% confirms results of previous
studies of patients with multitrauma. Glucan decreased pneumonia and sepsis to a
significantly lower level of 9.5%....The mortality rate related to infection decreased from 30.0
to 4.8%. The lower number of instances of pneumonia and sepsis....decreased the period of
time in the intensive care and the hospital, with a global reduction of 40% on hospital cost."
INTERLEUKIN - INT J IMMUNOPHARMACOL, 1987, 9:3, 261-7; "The study demonstrates
that; (1) glucan will enhance IL-1 and IL-2 production and (2) elevations in lymphokine
production can be maintained up to 12 days post-glucan"
PARASITES - TULANE UNIVERSITY (USA); "Trypanosoma cruzi, the causative agent of
Chagas' disease, infects humans and animals..... Glucan significantly (P less than 0.05)
increased survival rate as denoted by 60%...."
PNEUMONIA - HOSPITAL ARTHUR RIBEIRO DE SABOYA (Brazil); "The mortality rate
26
related to infection was 30.0 percent in patients in the control group and 4.8 percent in the
group treated with glucan...."
RADIATION - RADIOPROTECTIVE EFFECT; "These results suggest that early after
irradiation glucan may mediate its radioprotection by enhancing resistance to microbial
invasion via mechanisms not necessarily predicated on hemopoietic recovery. In addition,
preliminary evidence suggests that glucan can also function as an effective free-radical
scavenger."
RADIATION SURVIVABILITY- ARMED FORCES RADIOBIOLOGY RESEARCH
INSTITUTE (USA); "Immunomodulators, either microbial agents (e.g. glucan, TDM) or
recombinant cytokines (e.g. Interleukin-1, colony-stimulating factor), can enhance
hematopoietic and functional cell recovery after irradiation."
RADIATION SURVIVABILITY; Abstract: "Glucan, a beta-1, 3 polyglucose, was administered
to mice either 1h before or 1h after a 650 rad exposure to cobalt-60 radiation. Compared to
radiation controls, glucan-treated mice consistently exhibited a more rapid recovery of
pluripotent stem cells and committed granulocyte, macrophage and erythroid progenitor
cells. This may partially explain the mechanism by which glucan also enhances survival in
otherwise lethally irradiated mice."
STAPHYLOCOCCAL WOUND INFECTION - VANDERBILT UNIVERSITY SCHOOL OF
MEDICINE (USA); "We conclude that PGG Glucan reduces the risk of staphylococcal
abscess formation."
STRESS, PHYSICAL, OR EMOTIONAL - TOWNSEND LETTER FOR DOCTORS,
(1996);"The following list includes benefits from the use of Beta 1,3-glucan supplementation:
Professional and amateur athletes as well as people who work outdoors intensively. People
under physical or emotional stress"
TRAUMA PATIENTS - TULANE UNIVERSITY (USA); "total mortality rate was significantly
less in the glucan group (0% versus 29%) (p less then 0.05), the mortality rate from sepsis
was not statistically different (0% versus 17.6%). Glucan therapy significantly decreased
septic morbidity (9.5% versus 49%; p less than 0.05). Serum IL-1 had a greater increase in
glucan patients on day 3 after trauma (143.4 +/- 19.3% versus 78.6 +/- 11.7%; p less than
0.05),..."
WOUND HEALING - EAST TENNESSEE STATE UNIVERSITY (USA); "These data indicate
that macrophage modulation with glucan phosphate will increase tensile strength in
experimental colon and skin wounds."
27
ANNEX B
Beta Glucan Studies and Research
Abel, G. & Czop, J.K.; - "Stimulation of human monocyte beta-glucan receptors by glucan particles
induces production of TNF-alpha and IL-1 beta, "Int. J. Immunopharmacolol, 14: 1363-1373. 1992.*
Abel, G. & Czop, J.K., - "Activation of Human Monocyte GM-CSF and TNF-alpha. Production by
Particulate Yeast Glucan," International Congress for Infectious Diseases, Montreal, Canada
(abstract). 1990. * Dept of Medicine, Harvard Medical School, Boston, MA. Quote: "Beta-glucans are
pharmacological agents that rapidly enhance the host resistance to a variety of biologic insults through
mechanisms involving macrophage activation."
Adachi Y., Ohno N., Yacomae T.; - "Preparation and antigen specificity of an anti- (1-->3)-beta-Dglucan antibody," Biol Pharm bull 17: 1508-1512; 1994. *
Adachi Y., Ohno N., Yacomae T.; - "Inhibitory effect of beta-glucans on Zymosan-mediated hydrogen
peroxide production by murine peritoneal macrophages in vitro," Biol Pharm Bull, 16: 462-467; 1993.
Adachi Y., Ohno N., Ohsawa M., Oikawa S.,Yacomae T.; - "Macrophage activation in vitro by
chemically cross-linked (1--3)-beta-D-glucans," Chem Pharm Bull (Tokyo), 38:988-992 1990.
Laboratory of Immunopharmacology of Microbial Products, Tokyo College of Pharmacy, Japan. *
Ainsworth A.J., - "A beta-glucan inhibitable Zymosan receptor on channel catfish neutrophils," Vet
Immunol Immunopathol, 41: 141-152. 1994. *
Almdahl SM, Bogwald J, Hoffman J, Seljelid R; - "Treatment of experimental peritonitis in rats by
transfer of peritoneal mononuclear cells from rats injected with semisoluble aminated glucan." Acta
Chir Scand 153(9): 535-539, Sep 1987. Dept of Surgery, University Hospital, Tromso, Norway. *
Almdahl SM, Bogwald J, Hoffman J, Seljelid R; - "The effect of splenectomy on Escherichia coli
sepsis and its treatment with semisoluble aminated glucan," Scand J Gastroenterol 22(3): 261-267;
Apr 1987. *
Almdahl SM, Bogwald J, Hoffman J, Seljelid R Giercksky KE; - "Protection by aminated glucan in
experimental endogenous peritonitis," Eur Surg Res 19(2): 78-85, 1987. *
Almdahl SM, Seljelid R; - "Semisoluble animated glucan: long-term efficacy against an intraperitoneal
E. coli challenge and its effect on formation of abdominal adhesions," Res Exp Med (Berlin) 187(5):
369-377, 198 . *
Andaluz E., Guillen A., Larriba G.; - "Preliminary evidence for a glucan acceptor in the yeast
Candida albicans," Biochem J.; 240: 495-502. 1986.
Anti-free Radical Activity of Beta (1-3) glucan Molecule. Seporga Laboratories, Sophia Antipolis,
France. Research Report. 1990.
Aono R., Hammura M. et al; - "Isolation of extracellular 28- and 42-kilodalton beta-1-3-glucanases
and comparison of three beta-1, 3-glucanases produced by Bacillus circulans IAM1165," Appl.
Environ. Microbiol 61: 122-129.1995
Babineau, et al., - "A Phase II Multicenter, Double-Blind Randomized, Placebo-Controlled Study of
Three Dosages of an Immunomodulator (PGG-Glucan) in High Risk Surgical Patients", Arch. Surg.;
129:1204-1210. 1994. Dept of Surgery, Deaconess Hospital, Harvard Medical School, Boston MA. *
Babineau, et al., - "Randomized Phase I/II Trial of a Macrophage-Specific Immunomodulator (PGG-
28
Glucan) in High Risk Surgical Patients", Annals of Surgery; 220: (5): 601-609. 1994. Dept of Surgery,
Deaconess Hospital, Harvard Medical School, Boston MA. * Quote: "PGG-glucan is safe and appears
to be effective in further reduction of the morbidity and cost of major surgery."*
Bacon J., et al., - "The Glucan Components of the Cell Wall of Baker's Yeast (Saccharomyces
cerevisiae) Considered in Relation to its Ultrastructure," Chemical Abstracts, 71:109168c. 1991.
Ballou CE; - "The yeast cell wall and cell surface;" The Molecular Biology of the Yeast
Saccharomyces. Cold Spring Harbor Laboratories. New York. p 335; 1982.
Benach J.L., et al., - "Glucan as an adjuvant for a murine Babesia microti immunization trial,"
Infection and Immunity, 35(3): 947-951. 1982. Quote: "These observations demonstrate that glucan is
an effective adjuvant in enhancing immunity to murine babesiosis."*
Beta (1-3) glucan 1.3 Glucan Activity in Mice: Intraperitoneal and Oral Applications. Baylor College
of Medicine. Research Summary. 1989.
Beta (1-3) glucan: "I1-1 Cytokine Release after Oral Application in Mice". Baylor College of Medicine.
Research Report. 1994.
Bogwald J, Johnson E, Hoffman J, Seljelid R, - "Lysosomal Glycosidase in Mouse Peritoneal
Macrophages Stimulated in Vitro with Soluble and Insoluble Glucans". J. Leukocyte Biol.; 35: 357-371.
1984. *
Bogwald J, Johnson E, Seljelid R; - "The Cytotoxic Effect of Mouse Macrophages Stimulated in vitro
by a beta. 1,3-D-Glucan from Yeast Cell Walls". Scand. J. Immuol. 15: 297-304. 1982. Institute of Med
Bio, U of Tromso, Norway. Quote: " Macrophages stimulated by an insoluble beta 1-3-D-glucan from
yeast cell walls were able to destroy tumor cells as measured by the release of radioactive label from
prelabelled 14C-thymidine cells. Target cells were B-16 melanoma, P-815 mastocytoma, and the L929 cell line. A significant target cell killing by macrophages stimulated by glucan was observed after
72-96 h."
Bomford and Moreno, - "Mechanisms of the Anti-Tumor Effect of Glucans and Fructosans: A
Comparison with C. Parvum". Br. J. Cancer; 36:41-48. 1977.
Boone C, Sdicu A, Laroche M, Bussey H; - "Isolation from Candida albicans of a functional homolog
of the Saccharomyces cerevisiae KRE1 Gene, which is involved in cell wall beta-glucan synthesis," J
Bacteriol 173(21); 6859-6864, Nov 1991. *
Boone C., Sommer SS, Hensel A., Bussey H., - "Yeast KRE genes provide evidence for a pathway
of cell wall beta-glucan assembly," J Cell Biol; 110: 1833-1843. 1990.
Borriss, et al., - "Molecular cloning of a gene coding for thermostable beta-glucanase from Bacillus
macerns," J. Basic Microbiol; 28:3-10. 1988.
Borriss, et al., - "Expressions in Escherichia coli of a cloned beta-glucanase gene from Bacillus
Amyloliquefaciens," Appl. Microbiol. Biotechnol; 22:63-71. 1985.
Borriss, - Purification and characterization of an extracelluar beta-glucanase from Bacillus IMET B376
(1)), Z. Alg. Mikrobiologie; 21:7-17. 1981.
Borriss, et al., - "Beta-1, 3-1,4-glucanase in sporeforming microorganisms. V. The efficiency of betaglucanase in reducing the viscosity of wort", Zbl. Bakt II Abt. 136:324-329. 1981.
Bousquet M., Escoula L. et al; "Immunopharmacologic study in mice of 2 beta-1, 3, beta-1, 6
polysaccharides (Scleroglucan and PSAT) on the activation of macrophages and T lymphocytes," Ann
Rech Vet 20: 165-173. 1989. Station of Pharmacologie-Toxicologie, INRA, Toulouse, France. * Quote:
"...PSAT and scleroglucan favorably affect the non-specific host defense and cellular immune
29
response in mice."
Bousquet M., Escoula L., Pippy B, Besssieres MH, Chavant L, Seguela JP, "Enhancement of
Resistance of mice Toxoplasma gondi by 2 polysaccharides beta (1-3) glucan 1-3, beta (1-3) glucan
1-6 (PSAT and Scleroglucan)" Ann Parasitol Hum Comp., ^63 (6): 398-409. 1988. *
Bowers G., J. Patchen MLl, et al, "Glucan enhances survival in an intraabdominal infection model," J
Surg Res 47(2): 183-188; Aug 1989. *
Broach JR, Pringle JR and Jones EW; "The Molecular and Cellular Biology of the Yeast
Saccharomyces cerevisiae;" Genome Dynamics, Protein Synthesis, and Energetics; Cold Springs
Harbor Laboratory Press, Cold Spring Harbor, New York. 1991.
Browder W., Williams D., Pretus H., et al; Beneficial Effect of Enhanced Macrophage Function in the
Trauma Patients. Ann. Surg.; Vol 211: 605-613. 1990. Dept of Surg and Physiol, Tulane U Sch of
Med, LA and Istituto Di Chirurgia D'Urgenza, U of Torino, Torino, Italy. * Quote: "Previous studies have
demonstrated that glucan, a beta-1, 3-linked glucopyranose polymer, isolated from the inner cell wall
of Saccharomyces cerevisiae, is a potent macrophage stimulant and is beneficial in the therapy of
experimental bacterial, viral, and fungal diseases. Use of glucan in a murine model of hind-limb crush
injury decreased macrophage PGE2 release while stimulating bone marrow proliferation."
Browder Iw, Sherwood E., Williams D., Jones E., Mcnamee R., Diluzio N., "Protective effect of
glucan-enhanced macrophage function in experimental pancreatitis", Am J Surg.; 1153 (1): 25-33.
1987.
Browder W., et al., "Modification of Post-Operative C. albicans Sepsis by Glucan Immunostimulation,"
Int. J. Immunopharmac.; 6:19-26. 1984. Dept of Surg and Physiol, Tulane U Sch of Med, LA Quote:
"These observations suggest that Biologic Response Modifiers such as glucan may be effectively
employed in patients who are at risk for post-operative infections."*
Browder W., et al., "Protective Effect of Nonspecific Immunostimulation in Post Splenectomy Sepsis".
J. Surg. Res.; 35: 474-479. 1983. Dept of Surg and Physiol, Tulane U Sch of Med, LA * Quote: "This
study reports the use of glucan, a beta-1, 3-polyclucose, as a nonspecific immunostimulant for
postsplenectomy pneumococcal sepsis. ...Nonspecific immunostimulation appears to have significant
potential as a treatment strategy against postplenectomy infection."
Brown Jl, et al; "A mutational analysis of killer toxin resistance in Saccharomyces cerevisiae identifies
new genes involved in cell wall (1-->6)-beta-glucan synthesis," Genetics 133(4) 837-849, Apr 1993. *
Buddle BM, et al, "Protective effect of glucan against experimentally induced staphylococcal mastitis
in ewes." Vet Microbiol 16(1): 67-76, Jan 1988.
Bulone V., Fevre m.; "A 34-kilodalton polypeptide is associated with 1,3-beta-glucan synthase activity
from the fungus Saprolegnia monoica," FEMS Microbiol Lett: 140: 145-150, 1996.
Burgaleta C., Goide D. W.; Increased granulopoiesis and macrophage production in glucan-treated
mice; Chirigos MA, ed. Immune Modulation and Control of Neoplosia by Adjuvant Therapy. New York:
Raven Press, 195-219, 1978.
Burgaleta C., Territo M.C., Quan C.G., Goide D.W.; Glucan activated macrophages: functional
characteristics and surface morphology; J Reticuloendothel Soc 23: 195-204. 1978.
Burgaleta, C. and Golde, D.W.; "Effect of Glucan on Granulopoiesis and Macrophage Genesis in
Mice". Cancer Research; 37:1739-1742; Jun 1977. *
Cain J.A., Newman S.L., Ross G.D., "Role of complement receptor type three and serum opsonins in
the neutrophil response to yeast," Complement 4: 75-86.1987.
30
Campbell I And Duffus jh; eds., "Yeast." 1988.
Carrow, D.J.; "Beta-1, 3-glucan as a Primary Immune Activator," Townsend Letter; June 1996.
Cerenius L., Liang Z., Duvie B., et al, "Structure and biological activity of a 1,3 beta-D-glucanbinding protein in crustacean blood," J. Biol Chem 269: 29462-29467. 1994.
Cisreros RL, Gibson FC 3, Tzianabos AO; "Passive transfer of poly- (1-6)-beta-Glucotrisyl- (1-3)beta glucopyranose glucan protection against lethal infection in an animal model of intra-abdominal
sepsis," Infect Immun 64(6): 2201-2205, Jun 1996. Channing Laboratory, Brigham and Women's
Hospital, Boston, MA. *
Clark A.E., Stone B.A.; "Beta-glucan hydrolases from Aspergillus niger. Isolation of a beta- (1-4)glucan hydrolase and some properties of the beta- (1-3)-glucan-hydrolase components," Bichem J 96:
793-801. 1965.
Cook J. A., et al, "Protective Effect of Glucan against Visceral Leishmaniasis in Hamsters". Immun.;
37: 1261-1269. 1982.
Cook J. A., et al,, "Viscereal Leishmaniasis in Mice: Protective Effect of Glucan". J. Reticuloendothel;
Soc. 27: 567-573. 1980.
Cross CE, Bancroft GJ, "Ingestion of acapsular Cryptococcus neoformans occurs via mannose and
beta-glucan receptors, resulting in cytokine production and increased phagocytosis of the
encapsulated form." Infect Immun 63:2604-2611. 1995. Dept Clin Sci, London Sch of Hyg and Trop
Med, England.
Czop J.K., Kay J., Isolation and Characterization of B-glucan Receptors on Human Mononuclear
Phagocytes. J. Exp. Medicine; V.173: 1511-1520. 1991. (Copy available) Dept of Med, Harvard Med
Sch, Boston, MA. * Quote: "...human alveolar macrophages ...possess phagocytic receptors of
comparable ligand specificity for the Beta glucans commonly present in yeast and fungi. Pathogens
such as Candida and Aspergilli contain "yeast" glucan, cell wall components consisting of branched
homopolymers of Beta-D-glucose with 1,3 consecutive and 1,6-crosslinked chains and prototypic of
Saccharomyces cerevisiae."*
Czop J.K., Gurish M.F., Kadish J.l., Production and Isolation of Rabbit Anti-idiotypic Antibodies
Directed Against the Human Monocyte Receptor for Yeast B-glucans. Journal of Immunology;
145:995-1001. 1990. Dept of Med, Harvard Med Sch, Boston, MA. * Quote (p1): "Beta-Glucans with
1,3 and/or 1,6 linkages are the major structural components of yeast and fungi and are
pharmacological agents in animals...The cell wall glucans of S. cerevisiae consist of two structurally
distinct Beta-glucans: major components comprised of consecutively, 1,3-linked glucopyranosyl
residues with small numbers of 1,6-linked branches, and minor components with consecutive 1,6linkages and 1,3-branches."
Czop, J.K., Valiante N.M., Janusz M.J.; "Phagocytosis of particulate activators of the human
alternative complement pathway through monocyte beta-glucan receptors," Prog Clin Biol Res 297:
287-296; 1989. Dept of Med, Harvard Med S, Boston, MA. * Quote (p1): "Animal studies indicate that
beta-glucans with 1,3-and/or 1,6-linkages are active pharmacologic agents that rapidly confer
protection to a normal host against a variety of biological insults. The beta-glucan receptors provide a
mechanism by which a heightened state of host responsiveness is initiated."
Czop J.K., Puglisi A.V., Miorandi D.Z., Austen K.F.; "Pertubation of beta-glucan receptors on
human neutrophils initiates phagocytosis and leukotriene B4 production," J. Immunol 141: 3170-3176.
1988. *
Czop, Joyce K., "The Role of Beta-Glucan Receptors on Blood and tissue Leukocytes in
Phagocytosis and metabolic Activation". Pathology and Immunopathology Research; 5:286-296. 1986.
*
31
Czop J.K., Austen K.F., A B-glucan Inhibitable Receptor on Human Monocytes: Its Identity with the
Phagocytic Receptor for Particular Activators of the Alternative Complement Pathway. Journal of
Immunology 134: 1985; 2588-2593. 1985. *
Czop J.K., Austen K.F.; "Properties of glycans that activate the human alternative complement
pathway and interact with the human monocyte beta-glucan receptor," J Immunol 135: 3388-3393.
1985. *
Czop J.K., Austen K.F.; "A beta-glucan inhibitable receptor on human monocytes: its identity with the
phagocytic receptor for particulate activators of the alternative complement pathway," J Immunol
134(4): 2588-2593, Apr 1985. *
Czop J.K., Austen K.F.; "Generation of leukotrienes by human monocytes upon stimulation of their
beta-glucan receptor during phagocytosis," Proc Natl Acad Sci USA; 82: 2751-2755 1985. *
Daum T., Rohrbach M.S.; "Zymosan induces selective release of arachidonic acid from rabbit
alveolar macrophages via stimulation of a beta-glucan receptor," FEBS Lett 309: 119-122; 1992.
Deimann W, Fahimi HD, "The Appearance of Transition Forms Between Monocytes and Kupffer
Cells in the Liver of Rats Treated with Glucan," J Exp Med, p883-897, Apr 1979. * Dept of Anat, U of
Heidelberg, Germany.*
Delville, et al., entitled "Le-.beta-1, 3-Glucan et Autres Immunomodulateurs dans L'Unfection lepresis
Experimentale Chez La Souris". Acta Leprologica; 77/76: 273-281. 1979.
Deslanders, et al., "Triple-Helical Structure (1,3)-beta-D-Glucans". Macromolecules 13: 1466-1471.
1980.
Diluzio N.R., "Soluble phosphorylated glucan," U.S. Patent 487777, Issued Oct 31, 1989.
Diluzio N.R. (deceased), Williams D.L., Browder I.W.; Soluble phosphorylated glucan: methods and
compositions for treatment of neoplastic diseases; U.S. Patent 4818752; 1989.
Diluzio N.R.; Soluble phosphorylated glucan; U.S. Patent 4739046; 1988.
Diluzio N.R. and Williams D.L., " The Roll of Glucan in the Prevention and Modification of
Microparasitic Diseases;" in Chemical Regulation of Immunology in Veterinary Medicine, Alan R. Liss,
Inc.; pp. 443-456. 1984.
Diluzio N.R.,"Immunopharmacology of glucan: a broad-spectrum enhancer of host defense
mechanisms," Trends in Pharmacol. SCI., 4:344-347. 1983. Dept of Physiology, Tulane U, New
Orleans, LA. * Quote: (p347) "The broad spectrum of immunopharmacological activities of glucan
includes not only the modification of certain bacterial, fungal, viral and parasitic infections, but also
inhibition of tumor growth."
Diluzio N.R. Williams D.L. et al, "Comparative evaluation of the tumor inhibitory and antibacterial
activity of solubilized and particulate glucan," Recent Results Cancer Res 75:165-172. 1980. * Quote:
"Intravenous administration of soluble or particulate glucan resulted in significant reduction in the
growth of a syngeneic anaplastic mammary carcinoma and melanoma B16 and enhanced survival."
Diluzio NR, Williams DL; "Enhancement of host susceptibility to Staphylococcus aureus infection by
chronic ethanol ingestion-modification by glucan immunostimulation," Alcohol Clin Exp Res 4(3): 254260. Jul 1980. * Quote: "The administration of glucan significantly prolonged survival of S. Aureus
infected control and chronic ethanol mice."
Diluzio NR, Williams DL, et al, "Comparative tumor-inhibitory and anti-bacterial activity of soluble and
particulate glucan," Int J Cancer, 24(6): 773-779. Dec 1979. * Quote: "...these studies demonstrate
32
that a soluble glucan preparation exhibits significant anti-tumor and anti-staphylococcal activity."
Diluzio N.R. and Williams D.L., "Glucan-Induced Modification of the Increases Susceptibility of
Cyclophosphamide-Treated Mice to Staphylococcus aureus Infection". Cancer Immunol. Immunother.;
6: 73-79. 1979.
Diluzio NR, Williams DL, "Protective effect of glucan against systemic Staphylococcus aureus
septicemia in normal and leukemic mice," Infect Immun 20(3): 804-810. Jun 1978. * Dept of
Physiology, Tulane U, New Orleans, LA. * Quote: "These data denote that glucan enhances
nonspecific resistance to S. aureus sepsis, promotes survival during leukemic episodes, and
increases survival time of leukemic mice with experimentally induced staphylococcal infection."
Diluzio N.R., Williams D.L., Cook J.L., Hoffman E.O.; Protective effect of glucan in experimentally
induced Candidiasis; J Reticuloendothel Soc 53: 479-490, 1978.
Diluzio N.R., et al., "The Employment of Glucan and Glucan Activated Macrophages in the
Enhancement of Host Resistance to Malignancies in Experimental Animals," in The Macrophage in
Neoplasia; Academic Press, Inc. New York; pp. 181-198. 1976.
DiLuzio N.R., et al., "Evaluation of the Mechanism of Glucan-Induced Stimulation of the
Reticuloendothelial System". J. Reticuloendothelial Soc.; Soc.7: 731-742. 1970.
Di Renzo, L., Yefenof, E., Klein E., "The Function of human NK cells is enhanced by B-Glucan, a
ligand of CR3 (CD11b/CD18)". Eur. J. Immunol., 21:1755-1758. 1991.
Doita M, Rasmussen LT, Seljelid R, Lipsky PE, "Effect of soluble aminated beta-1, 3-D-polyglucose
on human monocytes: stimulation of cytokine and prostaglandin E2 production but not antigenpresenting function." J Leukoc Biol 49(4): 342-351. Apr 1991. *
Donzis B. A.; Substantially purified beta (1,3) finely ground yeast cell wall glucan composition with
dermatological and nutritional uses; U.S. Patent 5576015; 1996.
Donzis B.A.; Solubilized yeast glucan; U.S. Patent 5519009; 1996.
Donzis B.A.; Photoprotective composition containing yeast extract; U.S. Patent 5397773; 1995.
Donzis B.A.; Method of revitalizing skin by applying topically water insoluble glucan; U.S. Patent
5223491; 1993.
Duan X., Ackerly M. et al; "Evidence for involvement of beta-glucan-binding cell surface lectins."Cell
Immunol 157: 393-402; 1994.*
Duvic B., Soderhall K.; "Purification and partial characterization of a beta-1, 3-glucan-binding protein
membrane receptor from blood cells of the crayfish Pacifastacus leniusculus," Eur J. Biochem 207:
223-228; 1992.
Enhanced Healing of Decubitus Ulcers by Topical Application of Particulate Glucan. Tulane
University School of Medicine; Research Summary. 1984.
Bisu et al, "Studies on the Structure of Polysaccharides (Glucans and Fructans) Produced by
Cariogenic Streptococci and on an Enzyme Hydrolyzing the Insoluble Glucan I. Structural Studies of
Insoluble Glucan, Soluble Glucan, and Fructans," Chemical Abstracts; vol. 38:pp. 374-381. 1976.
Elstad MR, Parker C et al; "CD11b/CD18 integrin and a beta-glucan receptor act in concert to induce
the synthesis of platelet-activating factor by monocytes," J Immunol 152:220-230. 1994. Dept of Med,
Veterans Affairs Medical Center, Salt Lake City, UT. *
Engstad RE, Robertsen B, "Recognition of yeast cell wall glucan by Atlantic salmon (Salmo salar L.)
33
macrophages," Dev Comp Immunol 17:319-330. 1993. *
Felippe J., Silva M., Maciel F.M., et al., Infection prevention in patients with severe multiple trauma
with the immunomodulator beta (1-3) glucan 1-3 polyglucose (glucan). Surg. Gynecol Obstet., 177:
3833-388. 1993.
Fleet. G. H., et al., "Isolation and Composition of an Alkali-Soluble Glucan from the Cell Walls of
Saccharomyces cerevisiae," Chemical Abstracts; 85:89819z. 1976.
Fleet. G. H., et al.,"Isolation and Composition of an Alkali-Soluble Glucan from the Cell Walls of
Saccharomyces cerevisiae," Journal of General Microbiology; 94:180-192. 1976.
Franek J, Malina J, Kratka H, "Bacterial infection modulated by glucan: a search for the host defense
potentiation mechanisms," Folia Microbiol (Praha) 37(2): 146-152. 1992. *
Giaimis J., Lombard Y., et al; "Both mannose and beta-glucan receptors are involved in
phagocytosis of unopsonized, heat-killed Saccharomyces cerevisiae by murine macrophages," J.
Leukoc Biol 54: 564-571. 1993. *
Gillet et al., "Particulate beta 1-3 Glucan and Casual Prophylaxis of Mouse Malaria (Plasmodium
berghei)". In advances in Exper. Med. and Biology; vol 121A, Escobar and Friedman, eds. Plenum
Press, New York, pp. 307-313. 1980.
Glovsky MM, et al; "Effects of particulate beta-1, 3 glucan on human, rat, and guinea pig complement
activity," J. Reticuloendothel Soc. 33:401-413. 1983. * Quote: "Glucan administration is associated
with the modification of a variety of experimentally induced infectious disease states as well as the
inhibition of growth of implantable and spontaneous tumors."
Goldman R., "Characteristics of the B-glucan Receptor of Murine Macrophages". Exp.Cel. Res.;
174:481-490; 1988. *
Goldman R., "Induction of a beta-1, 3-D-Glucan Receptor in P388D1 Cells Treated with Retinoic Acid
of 1,25-dihydroxyvitamin D (3)," Immunology; 63:319-324. 1988.
Goto H., Yuasa K., Rylander R.; "(1-->3)-beta-D-glucan in indoor air, its measurement and in vitro
activity," Am J. Ind Med 25: 81-83.1994.
Hall MN and Linder P; "The Early Days of Yeast Genetics," Cold Spring Harbor Laboratory Press,
Cold Spring Harbor, New York. 1993.
Hara C., et al., "A Branched (1.fwdarw.3)-beta-D-Glucan From a Water Extract of Dictyophora
indusiata FISCH," Carb. Res.; 145:237-246. 1986.
Harada, et al., " Agricultural Biological, Growth and beta-Glucan 10C3K Production by a Mutant of
Alcaligenes faecalis var. myxogenes in Defined Medium"; vol. 30, No. 8, pp. 764-769. 1966.
Hartland RP, Emerson GW, Sullivan PA, "A secreted beta-glucan-branching enzyme from Candida
albicans," Pro R Soc Lond B Biol Sci, 246(1316): 155-160. Biochem Dept, U of Otago, Dunedin, New
Zealand. Nov 1991
Hassid, W.Z., Joslyn, M.A., McReady, R.M., "The Molecular Constitution of an Insoluble
Polysaccharide From Yeast Saccharomyces Cerevisae"; Journal of American Chemical Society, 1941;
63:295-298. 1941.
Hofemeister, "The beta-glucanase gene from Bacillus amyloliquefaciens shows extensive homology
with that of Bacillus subtilis," Gene; 49:177-187. 1986.
Holbrook J.A.C., Parker J.L.; Immunization against Leishmania donovani: glucan as an adjuvant with
34
killed promastigotes; Am J Trop Med Hyg 30: 762-768, 1981.
Holbrook T.W., et al., "Glucan-Enhanced Immunogenicity of Killed Erythrocylic Stages of
Plasmodium Benghei"; Infection and Immunity, 32, 542. 1981.
Honda S., et al, "Activation of the alternative pathway of complement by an antitumor (1----3)-beta-Dglucan from Alcaligenes faecalis var. myxogenes IFO 13140, and its lower molecular weight and
carboxymethylated derivatives,""Immunopharmacology 11:29-37. 1986. *
Inai et al., "Activation of the Alternative Complement Pathway by Water-Insoluble Glucans of
Streptococcus mutans: the Relation between Their Chemical Structures and Activating Potencies." J.
Immunol.; 117" 1256-1260. 1976.
Jamas S., Easson D., Ostroff G.R., DavidsonDd; "Method for producing soluble glucans," U.S.
Patent 5633369. Issued May 27, 1997. *
Jamas S., Easson D., Ostroff G.R.; "Glucan Preparation," U.S. Patent 5622939. Issued April 22,
1997. *
Jamas S., Easson D., Ostroff G.R.;"Glucan drug delivery system and adjuvant," U.S.Patent
5607677. Issued March 4, 1997. *
Jamas S., Easson D., Ostroff G.R.;"Use of aqueous soluble glucan preparations to stimulate platelet
production." U.S. Patent 5532223. Issued July 2, 1996. *
Jamas S., Easson D., Ostroff G.R.;"Use of neutral soluble glucan preparations to stimulate platelet
production." U.S. Patent 5488040. Issued January 30, 1996. *
Jamas S., Easson D., Ostroff G.R.;"Method for producing neutral glucans for pharmaceutical
applications," U.S. Patent 5322841. Issued June 21, 1994. *<
James S., Chen Y-CJ, Von Der Osten C.H., et al., "Spectral analysis of glucan produced by wildtype and mutant Saccharomyces cerevesiae". Carbohydr. Polym., 13:207-219. 1990.
Janusz M.J., Austen K.F., Czop J.K.; "Isolation of a Yeast Heptaglucoside that Inhibits Monocyte
Phagocytosis of Zymosan Particles". The Journal of Immunology; 142:959-965. 1989. Dept of Med,
Harvard Med Sch, Boston, MA. * Quote: "Beta-Glucans with 1, 3-and 1, 6 glycosidic linkages are the
major structural components of yeast and fungal cell walls and are active pharmacologic agents in
host defense systems of plants and animals.... The administration of particulate glucans from S.
cerevisiae to laboratory animals induces host resistance to a variety of lethal pathogens by
mechanisms involving macrophage stimulation. In vitro studies reveal that bone marrow-derived
mouse macrophages and human peripheral blood monocytes possess Beta-glucan receptors that
mediate phagocytosis of glucan particles and induce release of proinflammatory mediators..."
Janusz M.J., Austen K.F., Czop J.K.; Phagocytosis of Heat-killed Blastophores of Candida Albicans
by Human Monocyte B-glucan Receptors. Immunology; 65:181-185. 1988. *
Janusz M.J., Austen K.F., Czop J.K.; "Lysosomal enzyme release from human monocytes by
particulate activators is mediated by beta-glucan inhibitable receptors," J. Immunol 138: 3897-3901.
1987. *
Janusz M.J., et al, "Isolation of Soluble Yeast beta-Glucan that Inhibit Human Monocyte Phagocytosis
Mediated by beta-Glucan Receptors," L. Immunol; 137:3270-3276. 1986. *
Jiang B., Sheraton J., et al; "CWH41 encodes a novel endoplasmic reticulum membrane Nglycoprotein involved in beta 1, 6-glucan assembly," J. Bacteriol 178: 1162-1171. 1996.
Jones EW, Broach JR and Pringle JR. ;"The Molecular and Cellular Biology of the Yeast
35
Saccharomyces cerevisiae;" Gene Expression; Cold Springs Harbor Laboratory Press, Cold Spring
Harbor, New York. 1992.
Jorgensen J.B., Robertsen B.; "Yeast beta-glucan stimulates respiratory burst activity of Atlantic
salmon (Salmo salar L.) macrophages," Dev Comp Immunol 19: 43-57. 1995. *
Jorgensen J.B., "Quantification of high molecular weight (1-3)(1-4)-beta-D-glucan using calcofluor
complex formation and flow injection analysis. I. Analytical principle and its standardization," Carlsberg
Res. Commun. (1988); 53:277-285. 1988.
Kadish, J.L., Choi C.C., Czop J.K.; "Phagocytosis of unopsonized zymosan particles by trypsinsensitive and beta-glucan-inhibitable receptors on bone marrow-derived murine macrophages,"
Immunol Res 5: 129-138. 1986. *
Kan V.L., Bennett J.E.; "Beta 1, 4-oligoglucosides inhibit the binding of Aspergillus fumigatus conidia
to human monocytes," J Infect Dis 163: 1154-1156. 1991.
Kaplan J.; "Acceleration of Wound Healing by a Live Yeast Cell Derivative". Archives and Surgery",
Sep. 1984; 119:1005-1008. 1984.
Kapteyn J.C., Montijn R.C., et al; "Retention of Saccharomyces cerevisiae cell wall proteins through
a phosphodiester-linked beta-1, 3/beta-1, 6-glucan heteropolymer," Glycobiology 6: 337-345. 1996. *
Institute of Molecular Cell Biology, U of Amsterdam, The Netherlands.
Kapteyn J.C., Montijn R.C., et al; "Covalent association of beta-1, 3-glucan with beta-1, 6glucosylated mannoprotein in cell walls of Candida albicans," J Bacteriol 177: 3788-3792. 1995. *
Kapteyn J.C., Montijn R.C., et al; "Glucosylation of cell wall proteins in regenerating spheroplasts of
Candida albicans," FEMS Microbiol Letter 128: 271-277. 1995. *
Kapteyn J.C., Montijn R.C., et al; "Identification of beta-1, 6-glucosylated cell wall proteins in yeast
and hyphal forms of Candida albicans," Euro J Cell Biol 65: 402-407. 1994. *
Kasahara S., Ben Inoue S., Mio T., Yamada T., Nakajima T., Ichishima E., Furuichi Y., Yamada
H., "Involvement of cell wall beta (1-3) glucan-glucan in the action of HM-1 killer toxin", FEBS Lett 348
(1): 27-32. 1994.
Kasai, S., Fujimoto S., Nitta K., Baba H., Kunimoto T., "Antitumor activity of polymorphonuclear
leukocytes activated by a B-1, 3-D-glucan". J. Pharmacobiodyn. 14:519-525. Medline.
Kashkina Ma., Freidlin IS., "Macrophage activation by polysaccharides from yeast-like fungi", Biull
Eskp Biol. Med 89 (4): 439-441. 1980.
Kay J., Czop J.K., ""Enhancement of human monocyte beta-glucan receptors by glucocorticoids,"
Immunology 81: 96-102. 1994. *
Kimura, et aL., "In Vitro Activation of Human Adherent Cells by a Glucan, Schizophyllan". J.
Reticuloendothel.; Soc. 34: 1-11. 1983.
Kohl, et al., "Inhibition of Human Monocyte-Macrophage and Lymphocyte Cytotoxicity to Herpes
simplex Cells by Glucan". J. Immunol. Methods; 29: 361-368. 1979. * Quote: "Particulate, cellassociated glucan irreversibly inhibited MP antibody-dependent cellular cytotoxicity (ADCC)."
Kokoshis P.L., Williams D.L., Cook J.A., Di Luzio N.R.; Increased resistance to Staphylococcus
aureus infection and enhancement in serum lysozyme activity by glucan. Science 199: 1340-1342,
1978. * Quote: "These studies indicate that glucan confers an enhanced state of host defense against
bacterial infections."
36
Konopski Z., Seljelid R., Eskeland T.; "Interferon-gamma inhibits endocytosis of soluble animated
beta-1, 3-D-glucan and neutral red in mouse peritoneal macrophages," J Interferon Cytokine Res
15(7): 597-603. Jul 1995. * Dept of Exper Path and Anat, U of Tromso, Norway.
Konopski Z., Seljelid R., Eskeland T.; "IFN-gamma inhibits internalization of soluble aminated beta1-3-D-glucan by macrophages and thereby down-regulates the glucan induced release of TNF-alpha
and IL-1 beta," Scand J. Immunol 40: 57-63. 1994. *
Konopski Z., Seljelid R., Eskeland T.; "A novel immunomodulator soluble aminated beta-1, 3-Dglucan: binding characteristics to mouse peritoneal macrophages," Biochem Biophys Acta 1221(1):
61-65. Mar 1994. *
Konopski Z., Seljelid R., Eskeland T.; "Cytokines and PGE2 modulate the phagocytic function of the
beta-glucan receptor in macrophages," Scand J. Immunol 37: 587-592. 1993. *
Konopski, Z., et al., "Phagocytosis of beta-1, 3-D-Glucan-Derivatized Microbeads by Mouse
Peritoneal Macrophages Involves Three Different Receptors," Scand. J. Immunol.; 33:297-306. 1991.
*
Kopecka M.; "Electron microscopic study of purified polysaccharide components glucans and mannan
of the cell walls in the yeast Saccharomyces cerevisiae," J Basic Microbiol 25: 161-174. 1985. *
Lahnborg G., Hedstrom K.G., Nord C.E.; "The Effect of Glucan - A Host Resistance Activator and
Ampicillin on Experimental Intraabdominal Sepsis". Journal of Reticuloendothelial Society. 32: 347353. 1982. * Quote: "It is concluded that glucan, in combination with Ampicillin, has a significant effect
on the survival rate of rats with induced peritonitis, probably by enhancing the activities of the
reticuloendothelial system, an important part of the total host resistance."
Lahnborg, et al., "Glucan-Induced Enhancement of Host Resistance in Experimental Intraabdominal
Sepsis". Eur. Surg. Res.; 401-408. 1982. *
Larm O., Seljelid R., "Water-soluble aminated beta-1, 3-bount D-glucan and composition containing
same," U.S. Patent 4707471; Issued Nov 17, 1987.
Leibovich S.J., et al., "Promotion of Wound Repair in Mice by Application of Glucan". J.
Reticuloendothel, Soc. 27: 1-11. 1980.
Lejeune FJ., Vercammen-Granfjean A., Mendes Da Costa P., Bron D., Defleur V., "Suppressor cell
induction and reticuloendothelial cells activation produced in the mouse by beta(1-3)glucan 1-3
glucan", Adv. Exp. Med. Biol. 121 (A): 235-244. 1979. *
Lotzova and Gutterman, "Effect of Glucan on Natural Killer (NK) Cells: Further Comparison between
NK Cell and Bone Marrow Effector Cell Activities". J. Immunol., 123: 607-611. 1979.
Mahauthaman R, Howell CJ, Spur BW, Youlten LJ, Clark TJ, Lessof MH and Lee TH; "The
generation and cellular distribution of leukotriene C4 in human eosinophils stimulated by unopsonized
zymosan and glucan particles;" J Allergy Clin Immunol 81:696. 1988.
Manners, D.J., Masson, A.J. & Patterson, J.C.: "Heterogeneity of Glucan Preparations from the
Walls of Various Yeasts". J. of Gen Micro.; 411-417. 1974.
Manners, D.J., et al., "The Structure of a beta- (1.fwdarw.3)-D-Glucan from Yeast Cell Walls,"
BiochemJ.; 135: 19-30. 1973.
Mansell P.W.A., Rowden G., Hammer C.; Clinical experiences with the use of glucan. Chirigos MA,
ed.; Immune Modulation and Control of Neoplasia by Adjuvant Therapy. Raven Press, New York 255280; 1978.
37
Mansell P.W.A., et al., Activation of the Alternative Complement Pathway by Water-Insoluble Glucans
of Streptococcus mutans: the Relation Between Their Chemical Structures and Activating Potencies".
Macrophage-Mediated Destruction of Human Malignant Cells in Vitro; Inai et al., J. Immunol (1976);
1256-1260. 1976
Mansell P.W.A., Ichinose H., Reed R.J., Krements E.T., McNamee R.B., Di Luzio N.R.;
"Macrophage-mediated Destruction of Human Malignant Cells in Vitro". Journal of National Cancer
Institute; 54: 571-580. 1975. Quote: "The initial 9 patients studied had malignant carcinoma of the
breast. Control and experimental lesions were injected; subsequently biopsies were performed at
varying intervals for histologic evaluation. Always when glucan or glucan and RF fractions were
administered intra-lesionally; the size of the lesion was strikingly reduced in as short a period as 5
days. ...In small lesions, resolution was complete, whereas in large lesions, resolutions was partial."
Marchetti M., Pisani S., Pietropaolo V., Seganti L., Nicoletti R., Degener A., Orsi N., "Antiviral
effect of a polysaccharide from Sclerotium glucanicum towards herpes simplex virus type 1 infection",
Planta. Med. 62 (4): 303-307. 1996.
Mashiba, et Al., "In Vitro Activation of Human Adherent Cells by a Glucan Schlzophillan". Japan J.
Exp. Med; 53: 195-198. 1983.
Meira, D.A., et al; The Use of Glucan as Immunostimulant in the Treatment of
Paracoccidioidomycosis; Am J. Trop Med Hyg 55(5), 496-503; 1996. Dept of Trop Dis, Dept of
Microbio, State U of Sao Paulo, Brazil. Quote: "...glucan enhances the immune response through
stimulation of macrophages by increasing their number, size, and function, stimulates secretion of
lysozyme and TNF by activated macrophages, increases the phagocytosis of antigens, activates the
formation of granulocyte and monocyte colonies, and factors increased activity of T and B
lymphocytes, as well as complement activation. "
Mitsutake K, et al; "Enolase Antigen, Mannan Antigen, Cand-Tec Antigen, and (-Glucan in Patients
with Candidemia," J of Clin MicroB, p1918-1921, 1137; 1996. Copyrighted. Sec Dept of Int Med,
Nagasaki U Sch of Med, Nagasaki, Japan. *
Miura N.N., Ohno N., Adachi Y., Yadomae T.; "Characterization of sodium hypochlorite degradation
of Beta-glucan in relation to its metabolism in vivo," Chem Pharm Bulletin (Tokyo) 44: 2137-2141.
1996. *
Miyazaki, T., et al., "Structural Examination of Antitumor, Water-Soluble Glucans from Grifora
umbrellata by Use of Four Types of Glucanase," Carbohydrate Research; 65:235-243. 1978.
Modification of Experimental Viral Hepatitis by Glucan Induced Macrophage Activation".
Elesevier/North Holland Biomedical Press; pp. 363-368. 1980.
Morikawa K., Takeda, M., Yamazaki, M., and Mizuno D., "Induction of tumoricidal activity of
polymorphonuclear leukocytes by a linear B-1, 3-D-glucan and other immunomodulators in murine
cells". Cancer Res., 45: 1496-1501. (Medline).
Montijn RC et al, "Glucomannoproteins in the cell wall of Saccharomyces cerevisiae contain a novel
type of carbohydrate side chain," J Biol Chem, 269(30): 19338-19342: Inst of Molecular C Biol, U of
Amsterdam, The Netherlands. Jul 1994.
Mortimer RK, Contopoulou CR, King JS, "Genetic and physical maps of Saccharomyces
cerevisiae," Edition 11. Yeast 8:817-902. 1992.
Muller A., Rice P.J., Ensley H.E., et al; "Receptor binding and internalization of a water-soluble (1->3)-beta-D-glucan biologic response modifier in two monocyte/macrophage cell lines," J.Immunol 156:
3418-3425. 1996. *
Murphy, "The DNA sequence of the gene and genetic control sites for the execration B. subtilis
38
enzyme beta-glucanase," Nucleic Acids Res.; 12:5355-5367. 1984.
Nemoto J., Ohno N., et al; Analysis of cytokine in mBNAs induced by the administration of a highly
branched (1-3)-B-D-glucan. OL-2. Biol. Pharm Bull. 17:948-54; 1994.
Niki L., Allbright L., "Composition and method to enhance the efficacy of a fish vaccine and to
stimulate the immune system of fish." (A method to stimulate the immune system by a beta (1-3)
glucan having a beta (1-3) glucan-1, 3-linked main chain with beta (1-3) glucan-1, 6-linked single
glucose side chains); U.S. Patent 5189028. Issued February 23, 1993.
Niskanen E.O., Burgaleta C., Cline M.J., Goide D.W.; Effect of glucan, a macrophage activator, on
murine haemopoietic cell proliferation in diffusion chambers in mice; Cancer Res 38: 1406-1409,
1978.
Norton MD, JA [Prof. of Surg, Chief of Endocrine and Oncologic Surgery]; "Editorial: Annals of
Surgery," Washington University School of Medicine, Nov 1994. Quote: "In a prospective, randomized
double-blind study, [Babineau, et.al.] demonstrate that the preoperative administration of PGG-glucan,
a substance derived from yeast that increases the microbial killing activity of leukocytes, can decrease
infectious complications in patients undergoing major surgical procedures...the preliminary results are
positive and should be interpreted as good news."
Nuyen and Stadtsbaeder, "Comparative Biological and Antitoxoplasmic Effects of Polysaccharides,
In Vitro". In Advances in Exper. Med. and Biology, vol. 121A Escobar and Friedman, eds. Plenum
Press, New York; pp. 255-266. 1980.
Ohno N., Terui T., Chiba N., Kurachi K., Adachi Y., Yadomae T.; "Resistance of highly branched
(1-->3)-beta-D-glucans to formolysis," Chem Pharm Bulletin (Tokyo) 43: 1057-1060. 1995. *
Olson E.J., Standing J.E, et al; "Fungal beta-glucan interacts with vitronectin and stimulates tumor
necrosis factor alpha release from macrophages," Infect Immun 64: 3548-3554. 1996.
Onderdonk, AB., et al., "Anti-Infective Effect of Poly-.beta.1-6 -Glucotrisyl-beta 1-3-Glucopyranose
Glucan In Vivo," Infec. Immun.; 60:1642-1647. 1992. Dept of Pathology, Channing Lab, Brigham and
Women's Hospital, Boston, MA. * Quote: "Mice challenged with Escherichia coli or Staphylococcus
aureus were protected against lethal peritonitis by the intravenous administration of 10 micrograms of
poly-beta 1-6-glucotriosyl-beta 1-3-glucopyranose (PGG) glucan per animal 4 to 6 h prior to bacterial
challenge."
Ostroff, G.R.; "Inhibition of infection-stimulated oral tissue destruction by beta (1-3) glucan (1,3)glucan," U.S. Patent 5622940. Issued April 22, 1997.>
Ostroff, et al., "Manipulation of Yeast Glucan Structure: Molecular Weight, Branch Frequency and
Branch Length". The Fermentor, 9(12)L51, American Cancer Society; Abstract No. 19.; Aug 1989.
Patchen M.L., D'Alesandro M.M., Brook I., Blakely W.F. Mcvittie T.J.; "Glucan: Mechanisms
Involved in Its 'Radioprotective' Effect". J Leuc Biol.; 42:95-105. 1987.
Pachen M.L. Macvittie TJ, "Comparative effects of soluble and particulate glucans on survival in
irradiated mice," J Biol Response Mod 5(1): 45-60. Feb 1986. * Experimental Hematology Dept,
Armed Forces Radiobiology Research Inst, Bethesda, MD. Quote: "Both glucan-P and glucan-F
enhanced the recovery of peripheral blood white cell numbers, platelet numbers, and hematocrit
values. In addition, both agents increased endogenous pluripotent hemopoietic stem cell numbers in
sublethally irradiated mice."
Pachen M.L., Macvittie T.J.; "Stimulated Hemopeiesis and Enhanced Survival Following Glucan
Treatment in Sublethally and Lethally Irradiated Mice". Int. J. Immunopharmac; 7: 923-932. 1985.
Patchen M.L., et al., J. Biol. Res. Mod.; 3:627-633. 1984. Patchen M.L., McVittie T.J.; Temporal
39
Response of Murine Pluripotent Stem Cells and Myeloid and Erythroid Progenitor Cells to Low-dose
Glucan Treatment. Acta Hemat; 70:281-288. 1983. Experimental Hematology Dept, Armed Forces
Radiobiology Research Insti, Bethesda, MD. * Quote: "Clearly, there are numerous possible uses for
an agent such as glucan, which is a potent stimulator of hemopoietic activity. Currently, we [U.S.
Armed Services] are using glucan to enhance hemopoietic proliferation in conjunction with
hemopoietic injury induced by radiation."
Patchen, M.L., Lotzova E.; Modulation of murine hemopoiesis by glucan; Exp Hermatol 8: 409-422,
1980.
Patent Abstracts of Japan; "Production of beta-1, 3-glucan" (24 May 1989) vol. 13, No. 224, (C-599)
{3572) & Japanese Patent Application No. 137297; 7 Feb. 1989.
Patent Abstracts of Japan; "Production of beta-1, 3-glucan by cell of genus Euglena," 15 Aug 1988,
vol. 12, No. 299, (C-520) {3146} & Japanese Patent Application No. 6371192, 31 Mar. 1988.
Paulik S., Svrcek S., et al; "The effect of fungal and yeast glucan and levamisole on the level of the
cellular immune response in vivo and leukocyte phagocytic activity in mice," Vet Med (Praha) 37: 675685. 1992. *
Petre, et al., "Purification and properties of an endo-beta-1, 4-glucanase from Clostridium
thermocellum," (abst.) 7-Enzymes, (1981); 95:145879q, Biochemie; 63:629-639. 1981.
Popisil, et al., "Glucan Induced Hemopoietic Recovery in Gamma-Irradiated Mice". Experientia; 38:
1232-1234. 1982.
Poutsiaka D.D., et al, "Cross-linking of the beta-glucan receptor on human monocytes results in
interleukin-1 receptor antagonist but not interleukin-1 production," Blood 82: 3695-3700; 1993. Dept of
Med, New England Med Ctr, Boston, MA. Quote: "Because of their differential effects on cytokine
production, beta-glucans may be used to therapeutic advantage in the diseases in which IL-1 is
implicated."*
Proctor J.W., Stiteler R.D., Yamamura Y., Mansell P.W., Winters R., "Effect of glucan and other
adjuvants on the clearance of radiolabeled tumor cells from mouse lungs", Cancer Treat. Rep. ^2 (11):
1873-1880. (1978).
Proctor and Yamamura; "Letters to the Editor: Effectiveness of Glucan in the Treatment of Human
Neoplasia". J. Nat'l Cancer Inst.; 61: 1179-1180. 1978.
Radioprotective Effect of Oral Administration of Beta (1-3) glucan" Research Report, Armed
Forces Radiobiology Research Institute, Bethesda, MD 1989.
Rasmussen, LT, Konopski Z, Oian P, Seljelid R; "Killing of Escherichia coli by mononuclear
phagocytes and neutrophils stimulated in vitro with beta-1, 3-D-polyglucose derivatives," Microbiol
Immunol 36(11):1173-1188. 1992. * Inst of Med Bio, U of Tromso, Norway.
Rasmussen, LT and Seljelid, R.: "Novel Immunomodulators With Pronounced In Vitro Effects
Caused by Stimulation of Cytokine Release", Journal of Cellular Biochemistry; 46:60-68. 1991. * Inst
of Med Bio, U of Tromso, Norway. Quote: "Beta-1, 3-D-polyglucose derivatives protect mice against
otherwise lethal bacterial infections."
Rasmussen LT, Seljelid R, "Dynamics of blood components and peritoneal fluid during treatment of
murine E. coli sepsis with beta-1, 3-D-polyglucose derivatives. I. Cells.," Scand J Immunol 32(4): 321331. Oct 1990. *
Rasmussen LT, Seljelid R, "Dynamics of blood components and peritoneal fluid during treatment of
murine E. coli sepsis with beta-1, 3-D-polyglucose derivatives. II. Interleukin 1, tumor necrosis factor,
prostaglandin E2 and leukotriene B4," Scand J Immunol 32(4): 333-340. Oct 1990. *
40
Rasmussen LT, Seljelid R, "The modulatory effect of lipoproteins on the release of interleukin 1 by
human peritoneal macrophages stimulated with beta-1, 3-D-polyglucose derivatives." Apr 1989. *
Rasmussen LT, Seljelid R, "Production of prostaglandin E2 and interleukin 1 by mouse peritoneal
macrophages stimulated with beta-1, 3-D-glucan derivatized plastic beads," Scand J Immunol 26(6):
731-736. Dec 1987. *
Rasmussen, LT, Fandrem. Jr., and Seljelid R., "Dynamics of Blood Components and Peritoneal
Fluid During Treatment of Murine E. Coli Sepsis with beta-1, 3-D-polyglucose Derivatives"; Scand. J
63:73-80 Immunol. 1985.
Ray P.M.; "Cooperative action of beta-glucan synthetase..." Biochim Biophys Acta 629: 431-444.
1980.
Rios-Hernandez M., Dos-Santos N.J., Silvia-Cardosa, Belle-Garciga J.l., Peddrosa M.,
"Immunopharmacological studies of beta (1-3) glucan-1, 3-glucan", Arch. Med. Res. 25 (2): 179-180.
1994. *
Robertsen B., Engstad R.E., Jorgensen J.B.; "Beta (1-3) glucan-glucans as Immunostimulants in
Fish". Modulators of Fish Immune Responses; V.1.; 1994.
Roemer T, et al; "Characterization of the yeast (1-->6)-beta-glucan biosynthetic components, Kre6p
andSkn1p, and genetic interactions between the PKC1 pathway and the extracellular matrix
assembly," J Cell Bio 127(2): 567-579. Oct 1994. *
Roemer T, Delaney S, Bussey H; "SKN1 and KRE6 define a pair of functional homologs encoding
putative membrane proteins involved in beta-glucan synthesis," Mol Cell Biol 13(7): 4039-4048. Biol
Dept, McGill U, Montreal, Quebec, Canada, Jul 1993. *
Roemer T, Bussey H; "Yeast beta-glucan synthesis: KRE6 encodes a predicted type II membrane
protein required for glucan synthesis in vitro and for glucan synthase activity in vitro," Proc Natl Acad
Sci USA, 88(24): 11295-11299. Dec 1991.
Ross, G.D., Cain J.A., Myones B.L. et al; "Specificity of membrane complement receptor type three
(CR3) for beta-glucans," Complement 4: 61-74. 1987.
Sandula J., Machova E., Hribalova V.; "Mitogenic activity of particulate yeast beta- (1-->3)-D-glucan
and its water soluble derivatives," Int J Biol Macromol 17: 323-326. 1995. *
Sanjuan R., Zueco J, Stock R, Font De Mora J, Sentandreu R; "Identification of glucanmannoprotein complexes in the cell wall of Candida albicans using a monoclonal antibody that reacts
with a (1,6)-beta-glucan epitope," Microbiology, 141(Pt 7): 1545-1551; Dept de Microbiol, Facultat de
Farmacia, U. de Valencia, Burjassot, Spain. Jul 1995.
Sarko, et al.; "Multiple-Helical Glucans". Biochem. Soc. Trans.; 11: 139-142. 1983.
Sarko, et al., "Antitumor Activity of Tetrahydro-2-furnal-antetrahydro-2-pyranyl-Glucans Obtained by
Chemical Modification of (1,3)-beta D-Glucan from Alcaligenes faecalis var. myxogenes IFO 13140
and its Lower Molecular Weight Glucans" Cancer Treat Rep. (1983) Rep. 67:275-280. 1983.
Sasaki, et al., "Dependence on Chain Length of Antitumor Activity of (1,3)-beta-D-Glucan from
Alcaligenes faecalis var. myxogenes IFO13140, and its Acid-degraded Products". Cancer Res; 38:
379-383. 1978.
Schimanski D.; "Cosmetic agent containing natural yeast cell contents," U.S. Patent 4540571; Issued
Sep 10, 1985.
41
Schwarz, et al., "Isolation of a Clostridium thermocellum gene encoding a thermostable beta-1, 3glucanase (laminarinase),"Chemical Abstracts, (1988); 108:217067k, Biotechnology letters; 10(4):
225-230. 1988.
Seljelid R, et al, "Evidence that tumor necrosis induced by aminated beta 1-3D polyglucose is
mediated by a concerted action of local and systemic cytokines," Scand J Immuno 30(6): 687-694.
Dec 1989. * Quote: "Aminated beta 1-3D polyglucose (AG) causes regression of Meth A sarcoma in
syngeneic mice when injected systemically on day 7 after tumor inoculation. AG does not concentrate
in the tumor, but distributes throughout the body. AG treatment causes release of large amounts of
interleukin 1 (IL-1) both in vivo [in the body] and in macrophage cultures in vitro [out of body]."
Seljelid R, "Tumor regression after treatment with aminated beta 1-3D polyglucose is initiated by
circulatory failure," Scand J Immunol 29(2): 181-192; Feb 1989. *
Seljelid R., ET AL., "The protective effect of beta 1-3D-glucan-derivatized plastic beads against
Escherichia coli infection in mice," Scand J. Immuno 25(1): 55-60. Jan 1987. * Quote: "Pretreatment
with beta-1, 3-D-glucan-derivatized plastic beads conferred strong protection against Escherichia coli
infection in mice."
Seljelid R, "A water-soluble aminated beta 1-3D-glucan derivative causes regression of solid tumors
in mice," Biosci Rep 6(9): 845-851. Sep 1986. * Quote: "When water-soluble aminated beta 1, 3-Dglucan (AG) was injected intravenously or intraperitoneally on day 7 of tumor growth, the tumors
underwent complete regression."
Seljelid R., et al., "In vivo activation of mouse macrophages with beta-1, 3-D-glucan-derivatized
plastic beads," Scand J Immunol 21(6): 601-605. Jun 1985. *
Seljelid R.,et al., "A Soluble beta-1, 3-Glucan Derivative Potentiates the Cytostatic and Cytolytic
Capacity of Mouse Peritoneal Macrophages in Vitro". Immunopharmacol; 7: 69-73. 1984. *
Shandula I., Kogan G., Masler L.; "Structure and various characteristics of yeast beta-D-glucans,"
Vopr Med Khim 36: 39-42. 1990.
Sherwood. ER, et al., "Soluble Glucan and Lymphokine-activated Killer (LAK) Cells in the Therapy of
Experimental Hepatic Metastases," Chemical Abstracts; 108:179752V. 1988.
Sherwood. ER, et al., "Enhancement of Interleukin-1 and Interleukin-2 Production by Soluble
Glucan," International Journal of Immunopharmacology.; 9:(3):261-267. 1987.
Shibata Y., "Enzymatic hydrolysis of glucans containing beta-1, 3-and beta-1, 6-linkages. 3.
Gibberella beta-1, 6-glucan 6-glucanohydrolase operative in the selective hydrolysis of beta-1, 3glucosidic linkages in Eisemia laminaran," J. Biochem (Tokyo) 75: 85-92. 1974.
Shiota M., Nakajima T., Satoh A., Shida M., Matsuda K.; "Comparison of beta-glucan structures in a
cell wall mutant of Saccharomyces cerevisiae and the wild type," J Biochem (Tokyo) 98: 1301-1307.
1985.
Smedsrod B, Seljelid R, "Fate of intravenously injected aminated beta (1----3) polyglucose
derivatized with 125I-tyraminyl cellobiose," Immunophar 21(3): 149-158. May 1991. *
Spiros J.; Method for immune system activation by administration of a beta (1-3) glucan which is
produced by Saccharomyces cerevisiae strain R4; U.S. Patent 5504079; 1996.
Spiros J.; Use of neutral soluble glucan preparations to stimulate platelet production; U.S. Patent
5488040; 1996.
Spiros J., Rha C., Sinskey AJ; "Glucan compositions and process for preparation thereof," U.S.
Patent 4810646; Issued Mar 7, 1989.
42
Stashenko, et al., "Reduction of Infection-Stimulated Periapical Bone Resorption by the Biological
Response Modifier PGG Glucan", J. Dent. Res.; 74(1): 323-330; 1995. * Dept of Cytokine Biology,
Forsyth Dental Ctr, Boston, MA. Quote: "PGG glucan-treated animals had significantly less infectionstimulated periapical bone resorption than control animals..."
Steadman R., Petersen M.M., et al; "Differential augmentation by recombinant human tumor necrosis
factor-alpha of neutrophil responses to particulate zymosan and glucan," J. Immunol 144: 2712-2718.
1990. *
Stewart, et al., "Preliminary Observations on the Effect of Glucan in Combination with Radiation and
Chemotherapy in Four Murine Tumors", Cancer Treat. Prep.; 62: 1867-72. 1978.
Surarit R., Gopal P.K., Shephard M.G.; "Evidence for a glycosidic linkage between chitin and glucan
in the cell wall of Candida albicans," J. Gen Microbiol 134: 1723-1730. 1988.
Suzuki T., Ohno N., Adachi Y., Yadomae T., "Serum components induce beta-D-glucan-inhibitable
uptake of zymosan particles by murine peritoneal macrophages," Biol Pharm Bull: 16: 223-227. 1993.
*
Suzuki T., Ohno N., et al, "Activation of the complement system by (1--3)-beta-D-glucans having
different degrees of branching and different ultrastructures," J. Pharmacobiodyn 15: 277-285. 1992. *
Suzuki, Iwao, Tanaka, Hideki, Konoshita, Akira, Oikawa, Shozo, Osawa, Masumi and Yadomae.
"Effects of Orally Administered beta-Glucan on Macrophage Function in Mice". Toshiro, Journal of
Immunopharmac; vol. 12, No. 6, pp. 675-684. 1990.
Sveinbjornsson B, Seljelid R, "Aminated beta-1, 3-D-polyglucose activates salmon pronephros
macrophages in vitro," Vet Immunol Immunopathos 41(1-2): 113-123. May 1994.
Szabo T., Kadish J.L., Czop J.K.; "Biochemical properties of the ligand-binding 20-kDa of the betaglucan receptors on the human mononuclear phagocytes," J. Biol Chem 270: 2145-2151. 1995. *
Tanaka S., Aketagawa J., et al, "Inhibition of high molecular weight (1-->3)-beta-D-glucan-dependent
activation of a limulus coagulation factor G by laminaran oligosaccharides and curdlan degradation
products," Carbohydr Res 244: 115-127; 1993. *
Tanaka, Immunopharmac. and Immunotoxi.; 14:403-420. 1992. Tapper H., Sundler R.; "Glucan
receptor and zymosan-induced lysosomal enzyme secretion in macrophages," Biochem J. 306: 829835. 1995. *
Thompson I.M., Spence C.R. Lamn DL., Diluzio N.R., "Immunochemotherapy of bladder carcinoma
with glucan and cyclophosphamide", Am. J. Med. Sci. 294 (5): 294-300. 1987. *
Thornqvist P.O., Hohansson M.W., Soderhall K.; "Opsonic activity of cell adhesion proteins and
beta-1-3-glucan binding proteins from two crustaceans," Dev Comp Immunol 18: 3-12; 1994.
Thornton B.P., Vetvicka V., Pitman M., Goldman R.C., Ross G.D.; "Analysis of the sugar specificity
and molecular location of the beta-glucan-binding lectin site of the complement receptor type 3
D11b/CD18)," J. Immunol 156: 1235-1246. 1996.
Tikhomirov, et al, "Endo-1, 4-beta-glucanases of the anaerobic bacterium Clostridium thermocellum
st. No. 3 with high heat stability," Chemical Abstracts; 110:168879g.; 1989.
Todd, R.F.; "The Continuing Saga of Complement Receptor Type 3 (CR3)," J. Clin Invest.: Vol 98, 12. 1996. Div of Hematology/Oncology Dept of Int. Med, U of Michigan Med Ctr.* Quote: (p2) "In certain
controlled clinical trials, the increased survival of patients receiving these immunostimulatoryBetaglucans has been reported."
43
Tomos et al., "A protein-glucan intermediate during paramylon synthesis" Biochem. J.; 174:283-290.
1978.
Tong, D.W., Barnetson R.S.; B-1, 3-D-glucan gel in the treatment of solar keratoses; Australasian J
of Dermatology, 37: 137-138, 1996. * Dept of Dermat, Royal Prince Alfred Hosp, Camperdown,
Australia.
Truscheit E., Bierling R., Schlumberger H., Oettgen H.; "Process for the preparation of
immunopotentiating agents from components of yeast cell wall material; " U.S. Patent 4138479;
Issued Feb 6, 1979. *
Tsujinaka T., Yokota M.K.; Modification of septic processes by B-glucan administration. Eur Surg
Res; 22:540-546, 1990. *
Tzianabos AO, Cisneros RL; "Prophylaxis with the immunomodulator PGG glucan enhances
antibiotic efficacy in rats infected with antibiotic-resistant bacteria, "Ann NY Acad Sci 797: 285-287;
Oct 1996. * Quote: "Results of these studies demonstrated that prophylaxis with PGG glucan in
combination with antibiotics provided enhanced protection against lethal challenge with Escherichia
coli or Staphylococcus aureus as compared with the use of antibiotics alone."
Uchida, A.; "Method for treatment of chronic fatigue syndrome, "U.S. Patent 5424300 (A method for
the treatment of chronic fatigue syndrome, comprising administering a polysaccharide which further
contains a beta (1-3) glucan-1, 3/1, 6-glucoside bond). Issued June 13, 1995.
Van Der Vaart J.M., et al, and "The retention mechanism of cell wall proteins in Saccharomyces
cerevisiae Wall-bound Cwp2p is beta-1, 6-glucosylated," Biochim Biophys Acta, 1291(3): 206-214.
Dept Molecular Cell Biol, Utrecht U., The Netherlands. Dec 1996.
Van Der Vaart J.M., et al, "The beta-1,6-glucan containing side-chain of cell wall proteins of
Saccharomyces cerevisiae is bound to the glucan core of the GPI moiety," FEMS Microbiol Lett 145:
401-407. 1996.
Vargas-Albores F., Jimenez-Vega, Soderhall K.; "A plasma protein isolated from brown shrimp
(Penaeus californiensis) which enhances the activation of prophenoloxidase system by beta-1, 3glucan," Dev Comp Immunol 20: 299-306. 1996.
Vetvicka V., Thornton B.P., Ross G.D.; "Soluble Beta-glucan Polysaccharide Binding to the Lectin
Site of Neutrophil or Natural Killer Cell Complement Receptor Type 3 (CD11b/CD18) Generates a
Primed State of the Receptor Capable of Mediating Cytotoxicity of iC3b-Opsonized Target Cells,"
Journal Clin Invest 98: 50-61. 1996. Div of Experimental Immuno and Immunopath, Dept Path, U of
Louisville, KY. * Quote: "This investigation showed that soluble CR3-specific polysaccharides such as
beta-glucan induced a primed state of CR3 that could trigger killing of iC3b-target cells that were
otherwise resistant to cytotoxicity."
Wessels J.G.; "A beta 1,6-glucan glucanohydrolase involved in hydrolysis of cell-wall glucan in
Schizophyllum commune," Biochem Biophys Acta 178: 191-193. 1969.
Williams D.L. ,Mueller A., Mueller P., Swails W., et al., "Randomized phase I/II trial of a
macrophage-specific immunomodulator (PGG-glucan) in high-risk surgical patients". Ann. Surg.;
220(5): 601-609. 1994.
Williams D.L., et al,"Development of a Water-Soluble, Sulfated (1.fwdarw.3)-beta-D-Glucan Biological
Response Modifier Derived from Saccharomyces cerevisiae," Carbohydrate Research. 247-257. 1992.
Williams D.L., et al, Development, Physicochemical Characterization and Preclinical Efficacy
Evaluation of a Water Soluble Glucan Sulfate Derived from Saccharomyces cerevisiae,"
Immunopharmacology; 22:139-156. 1991.
44
Williams D.L., Mcnamee R.B., Jones E.L., et al., "A method for the solubilization of a (1-2)-B-Dglucan isolated from Saccharomyces cerevisiae." Carbohydr Res.; 219: 203-213. 1991.
Williams D.L., Browder I. and Diluzio N.R., "Soluble phosphorylated glucan: methods and
compositions for wound healing," U.S. Patent 4975421, Issued Dec 4, 1990. Quote: "The soluble
phosphorylated glucans are useful for promoting the wound healing process. The soluble
phosphorylated glucans are also useful for prophylactic and therapeutic applications against
neoplastic, bacteria, viral, fungal and parasitic diseases."
Williams D.L., Browder I. and Diluzio N.R., "Methods and compositions for prophylactic and
therapeutic treatment of infections," U.S. Patent 4900722, Issued Feb 13, 1990. Quote: "The soluble
phosphorylated glucans are also useful for stimulating macrophage cells, either in vivo or in vitro, to
produce a cytotoxic/cyctostatic factor effective against cancer cells."
Williams D.L., Sherwood E.R., Browder I.W., McNamee R.B., Jones E.L., Di Luzio N.R.; Preclinical Safety Evaluation of Soluble Glucan. International Journal Immunopharmac. 1988; 10: 405411. 1988.
Williams D.L., et al; "Pre-clinical Safety Evaluation of Soluble Glucan", Int. J. Immunophamac. Vol.
10, No. 4: 405-414. 1988. * Dept of Phys, Tulane U Sch of Med, New Orleans, LA. *Quote: "Soluble
glucan, a beta-1, 3-linked glucopyranose biological response modifier, is effective in the therapy of
experimental neoplasia, infectious diseases and immune suppression."
Williams D.L., et al; "Therapeutic efficacy of glucan in a murine model of hepatic metastatic disease,"
Hepatology 5(2): 198-206. Mar 1985. *Quote: "...coincubation of particulate glucan with diverse
populations of normal or tumor cells in vitro indicated that glucan exerted a direct cytostatic effect on
sarcoma and melanoma cells and, in contrast, had a proliferative effect on normal spleen and bone
marrow cells."
Williams D., Browder IW and Diluzio N.R, "Immunotherapeutic modification of Escherichia coliinduced experimental peritonitis and bacteremia by glucan," Surgery 93(3): 448-454. Mar 1983. *
Quote: "These data denote that the intraperitoneal administration of glucan significantly modifies the
course of E. coli-induced peritonitis and bacteremia due, in part, to glucan-induced enhancement of
macrophage function."
Williams D.L. and Diluzio N.R.; "Modification of Experimental Viral Hepatitis by Glucan Induced
Macrophage Activation". In the Reticuloendothelial System and Pathogenesis of Liver Disease, Liehr
and Grun, eds. Elsevier/North Holland Biomedical Press; pp. 363-368. 1983.
Williams D.L. and Diluzio N.R.; "Glucan-Induced Modification of murine Viral Hepatitis". Science
(1980), 208: 67-69. 1980. *Quote: "Thus glucan is capable of increasing survival, inhibiting hepatic
necrosis, and maintaining an activated state of phagocytic activity in mice challenged with [mouse
hepatitis virus strain] MHV-A59."
Williams D.L., et al; "Protective Effect of Glucan in Experimentally Induced Candidiasis". J.
Reticuloendothel; Soc 23: 479-490. 1978.
Williams D.L, Diluzio NR, "Glucan induced modification of experimental Staphylococcus aureus
infection in normal, leukemic and immunosuppressed mice." Adv Exp Med Biol 121(A): 291-306.
1979*
Williams D.L, Diluzio NR, , Reticuloendothelial System and Pathogenesis of Liver Disease; Liehr and
Grun. eds. Solubilization of a (1-3_-B-D-glucan isolated from Saccharomyces cerevisiae. Carbohydr.
Res. 219: 203-213. 1991.
Wolk, M. and Danon, D.; "Promotion of Wound Healing by Yeast Glucan Evaluated on Single
Animals"; Medical Biology; 63:73-80. 1985. *
45
Wooles and Diluzio N.R.; "The Phagocytic and Proliferative Responses of the Reticuloendothelial
System Following Glucan Administration". J. Reticuloendothelial..; Soc. 1: 169-169. 1964.
Yoxhida M, et al, "Soluble (1-->3)-beta-D-glucan purified from Candida albicans: biologic effects and
distribution in blood and organs of rabbits," J Lab Clin Med 128(1): 103-114. Jul 1996. *Dept of Lab
Med, U of Cal Sch of Med, San Francisco, CA.
Yoshida H., Ochiai M., Ashida M.; "Beta-1, 3-glucan receptor and peptidoglycan receptor are present
as separate entities within insect prophenoloxidase activating system," Biochem Biophys Res
Commun 141: 1177-1184. 1986.
46
Scaricare