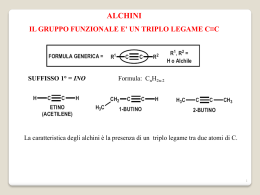
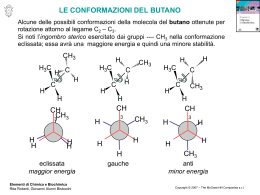
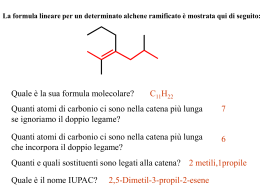
Fondamenti di chimica organica Janice Gorzynski Smith Copyright © 2009 – The McGraw-Hill Companies srl Soluzioni ai problemi proposti nel libro Capitolo 20 20.1 Il numero di legami C–N determina la classificazione come ammidi 1o, 2o, o 3o. 2° amide O H N N 2° amide N OCH3 O O 3° amide S H N N N OCH3 O 3° amide 20.2 All’aumentare della basicità di Z, aumenta la stabilità di RCOZ, a causa dell’aumento della stabilizzazione che deriva dalla risonanza. R O O C C R + Br Br O R C + Br The basicity of Z determines how much this structure contributes to the hybrid. Br– is less basic than –OH, so RCOBr is less stable than RCOOH. 20.3 O CH3 Cl CH3 NH2 CH3 H C O Cl CH3 O O C C NH2 H C Cl NH2 This resonance structure contributes little to the hybrid since Cl– is a weak base. Thus, the C–Cl bond has little double bond character, making it similar in length to the C–Cl bond in CH3Cl. This resonance structure contributes more to the hybrid since –NH2 is more basic. Thus, the C–N bond in HCONH2 has more double bond character, making it shorter than the C–N bond in CH3NH2. Fondamenti di chimica organica Janice Gorzynski Smith Copyright © 2009 – The McGraw-Hill Companies srl 20.4 a. (CH3CH2)2CH COCl O d. redraw H O C OCH2CH3 alkyl group = ethyl Cl 2-ethylbutanoyl chloride acyl group = formate 2-ethyl b. ethyl formate O C6H5COOCH3 e. CH3CH2 redraw C O O C benzoic propanoic anhydride O OCH3 methyl benzoate acyl group = propanoic alkyl group = methyl acyl group = benzoate c. CH3CH2CON(CH3)CH2CH3 redraw f. O acyl group = benzoic CN 3-ethylhexanenitrile 6 carbon chain = hexanenitrile N N-ethyl-N-methyl N-ethyl-N-methylpropanamide acyl group = propanamide 20.5 a. 5-methylheptanoyl chloride d. N-isobutyl-N-methylbutanamide g. sec-butyl 2-methylhexanoate O O O N Cl b. isopropyl propanoate O h. N-ethylhexanamide e. 3-methylpentanenitrile O CN c. acetic formic anhydride O f. o-cyanobenzoic acid O O H O O CH3 OH CN O N H Fondamenti di chimica organica Janice Gorzynski Smith Copyright © 2009 – The McGraw-Hill Companies srl 20.6 O Cl a. g. 2,2-dimethylpropanoyl chloride N O N-cyclohexylbenzamide H O b. h. cyclohexyl pentanoate O m-chlorobenzonitrile Cl CN c. O O i. Cl 3-phenylpropanoyl chloride isobutyl 2,2-dimethylpropanoate O d. 2-ethylhexanenitrile O j. C CN O Cl O e. cis-2-bromocyclohexanecarbonyl chloride Br O O k. N cyclohexanecarboxylic anhydride N,N-diethylcyclohexanecarboxamide O O f. cyclopentyl cyclohexanecarboxylate l. phenyl phenylacetate O O 20.7 a. propanoic anhydride O e. isopropyl formate H b. α-chlorobutyryl chloride O O O O i. benzoic propanoic anhydride O O O f. N-cyclopentylpentanamide O j. 3-methylhexanoyl chloride NH O Cl O Cl c. cyclohexyl propanoate k. octyl butanoate O d. cyclohexanecarboxamide h. vinyl acetate O O O NH2 O CN O C Cl g. 4-methylheptanenitrile O l. N,N-dibenzylformamide CH3 O H 20.8 Classificare i composti usando le regole della risposta 20.13. N Fondamenti di chimica organica Janice Gorzynski Smith Copyright © 2009 – The McGraw-Hill Companies srl O O a. OCH2CH2CH3 NH2 −NH 2 Cl −OCH CH CH 2 2 3 stongest base least reactive −Cl weakest base most reactive intermediate O b. O O O F3C O O ester least reactive anhydride intermediate O O O O CF3 anhydride with electron withdrawing F's most reactive O O c. OH − SH Cl − OH strongest base least reactive − SH intermediate Cl weakest base most reactive 20.9 Better leaving groups make acyl compounds more reactive. A has an electron withdrawing NO2 group, which stabilizes the negative charge of the leaving group, whereas B has an electron donating OCH3 group, which destabilizes the leaving group. O CH3 C O O CH3 NO2 A C O OCH3 B an electron withdrawing substituent an electron donating substituent O O O N O N O O O OCH3 OCH3 O one possible resonance structure leaving group from A one possible resonance structure leaving group from B Adjacent negative charges destabilize the leaving group. Delocalizing the negative charge on the NO2 stabilizes the leaving group making A more reactive than B. 20.10 resonance structures for the leaving group O O C N N R imidazolide Nu R C N Nu O N R C Nu N N N N N N N N N The leaving group is both resonance stabilized and aromatic (6 electrons), making it a much better leaving group than exists in a regular amide. N Fondamenti di chimica organica Janice Gorzynski Smith Copyright © 2009 – The McGraw-Hill Companies srl 20.11 Reaction as an acid: O CH3 O B C CH3 NH2 O C NH CH3 C CH3CH2 B NH2 NH CH3CH2 NH no resonance stabilization of the conjugate base These two resonance structures make the conjugate base more stable, and therefore CH3CONH2 a stonger acid. Reaction as a base: CH3 O O C C CH3 NH2 CH3CH2 This electron pair is localized on N. NH2 NH2 This electron pair is delocalized by resonance, making it less available for electron donation. Thus CH3CONH2 is a much weaker base. 20.12 Per disegnare i prodotti di queste reazioni di sostituzione nucleofila acilica, individuare il nucleofilo ed il gruppo uscente. Poi sostituire il gruppo uscente con il nucleofilo e disegnare un prodotto neutro. nucleophile O a. CH3 C O CH3OH Cl nucleophile CH3 C O + HCl OCH3 b. CH 3 leaving group C O NH3 OCH2CH3 CH3 C NH2 + HOCH2CH3 leaving group 20.13 Migliore è il gruppo uscente, più reattivo risulta il derivato dell'acido carbossilico. La base più debole è il miglior gruppo uscente. O O C C a. −NH NH2 stongest base least reactive b. − 20.14 C OCH3 −OCH 3 2 CH3CH2 O −Cl weakest base most reactive intermediate O O O C C C NHCH3 NHCH3 stongest base least reactive CH3CH2 − OH OH intermediate Cl CH3CH2 − O O C CH2CH3 OOCCH2CH3 weakest base most reactive I composti acilici più reattivi possono essere convertiti nei composti acilici meno reattivi. Fondamenti di chimica organica Janice Gorzynski Smith Copyright © 2009 – The McGraw-Hill Companies srl a. CH3COCl c. CH3COOH more reactive YES b. CH3CONHCH3 less reactive NO CH3COCl more reactive CH3COOCH3 NO less reactive CH3COOCH3 less reactive more reactive 20.15 CH3 O O O O C C C C O CH3 Cl3C O CCl3 acetic anhydride trichloroacetic anhydride The Cl atoms are electron withdrawing, which makes the conjugate base (the leaving group, CCl3COO–) weaker and more stable. 20.16 O a. O H2O O OH + N – H Cl pyridine Cl O b. CH3COO− + NH4+Cl– NH2 NH3 c. excess O O O + Cl– CH3 (CH3)2NH N(CH3)2 d. + (CH3)2NH2 Cl– excess 20.17 Il meccanismo ha 3 stadi: [1] attacco nucleofilo dell'O; [2] trasferimento di un protone; [3] eliminazione del gruppo uscente Cl− per formare il prodotto. O O Cl CH3OH O Cl OCH3 O CH3 O CH3 pyridine O Cl H H N N 20.18 O O O O H2O a. O OH NH3 HO c. pyridine O b. CH3OH O O HO NH2 NH4 O excess O CH3 O O (CH3)2NH d. excess O N(CH3)2 O (CH3)2NH2 Cl Fondamenti di chimica organica Janice Gorzynski Smith Copyright © 2009 – The McGraw-Hill Companies srl 20.19 La reazione di un acido carbossilico con cloruro di tionile lo converte in un cloruro dell'acido. O a. CH3CH2 O SOCl2 C OH CH3CH2 C O C b. Cl O [1] SOCl2 OH O [2] (CH3CH2)2NH (excess) C C Cl N(CH2CH3)2 20.20 COOH + CH3CH2OH a. H2SO4 C OCH2CH3 + H2O O O COOH b. + OH C H2SO4 + H2O O O COOH c. + C O + CH3OH O H2SO4 d. HO Na+ O NaOCH3 OH + H2O O 20.21 O O C CH318OH OH C 18 OCH3 + H2O 20.22 O HO C OH O H H−OSO3H C HO O HO OH H O – + HSO4 HO OH H O –OSO H 3 O O H O + H2SO4 H−OSO3H H2O + O HSO4– HO OH2 O + HSO4– 20.23 Fondamenti di chimica organica Janice Gorzynski Smith Copyright © 2009 – The McGraw-Hill Companies srl O a. O CH3NH2 + O O CH3NH2 O− NH3CH3 OH b. c. DCC O O CH3NH2 NHCH3 Δ OH NHCH3 OH + H2O 20.24 CH3CH2CH2CH2COCl NH3 H 2O a. CH3CH2CH2CH2COOH pyridine b. excess CH3CH2CH2CH2CONH2 (CH3CH2)2NH CH3CH2OH e. CH3CH2CH2CH2COOCH2CH3 pyridine O CH3COO− c. d. CH3CH2CH2CH2 O O excess CH3CH2CH2CH2CON(CH2CH3)2 C6H5NH2 CH3 f. excess CH3CH2CH2CH2CONHC6H5 20.25 O O O a. SOCl2 d. no reaction H2O 2 no reaction O O b. NaCl (CH3CH2)2NH OH e. excess N(CH2CH3)2 O O CH3OH c. O H2N(CH2CH3)2 OCH3 O O f. OH CH3CH2NH2 excess NHCH2CH3 O O 20.26 H3NCH2CH3 Fondamenti di chimica organica Janice Gorzynski Smith Copyright © 2009 – The McGraw-Hill Companies srl O OH O NaHCO3 a. + H2CO3 g. O Na O NaOH b. + H 2O h. c. O O OH [1] NaOH i. O SOCl2 OCH3 CH3OH – O Na O CH3OH H2SO4 [2] CH3COCl O Cl d. NaCl no reaction j. O CH3NH2 DCC NH3 e. O O NH4 k. NH2 l. O NH3 f. Δ NHCH3 [1] SOCl2 (1 equiv) O O O [2] CH3CH2CH2NH2 NHCH2CH2CH3 O [1] SOCl2 [2] (CH3)2CHOH OCH(CH3)2 20.27 O a. SOCl2 b. H3O+ O no reaction d. NH3 O O O H2O/–OH c. HO NH2 OH HO O HO e. CH3CH2NH2 NHCH2CH3 O 20.28 NH2 a. H3O+ CH2COOH b. H2O/–OH CH2COO O 20.29 a. H3O+ COOH d. [1] CH3CH2Li [2] H2O CN b. H2O/–OH COO [2] H2O C [2] H2O O [1] LiAlH4 CH2NH2 f. O H e. [1] DIBAL-H CH3 c. [1] CH3MgBr O [2] H2O HO Fondamenti di chimica organica Janice Gorzynski Smith Copyright © 2009 – The McGraw-Hill Companies srl 20.30 O COOH C SOCl2 a. pyridine OH g. [2] H2O/–OH h.C6H5CH2COOH + O [1] CH3CH2CH2MgBr C C6H5 [2] H2O d. (CH3)2CHCOOH + N C6H5 N H (excess) c. C6H5CN (CH3)2CH C NHCOCH3 O COOH O (CH3CO)2O + NH2 NHCOCH3 (excess) NH2 O + CH3COO– H3N O O f. HOOC OCH(CH3)CH2CH3 + O C6H5CH2CH2CH2COOH Δ j. O –OH H3O+ O k. H2O + H3O OH C6H5CH2CH2COOCH2CH3 l. OH H2O –OH C6H5CH2CH2COO– + HOCH2CH3 O O 20.31 R OH OH OH O H O H C C C C C OR' R R OR' OR' R OH product of step [1] R O H R OH C OH product of step [5] 20.32 O C H−OSO3H O CH3 O [2] CH3CH2CH2CH2NH2 [3] LiAlH4 C6H5CH2CH2NHCH2CH2CH2CH3 [4] H2O CH2CH2CH3 CH3 e. CH3CH2CH2CH2 [1] SOCl2 C6H5CH2CH2CH2CN i. O H2SO4 CH3CH2CHOH CH3CH2CH2CH2Br OH O b. C6H5COCl O [1] NaCN Cl OH OH H−OSO3H C O CH3 C OH + HSO4– OH O CH3 C OH H O CH3 C OH O CH3 OH H + HSO4– H2O + HSO4– O C O H O H + H2SO4 C OH HSO4– CH3OH Fondamenti di chimica organica Janice Gorzynski Smith Copyright © 2009 – The McGraw-Hill Companies srl 20.33 In queste condizioni l'estere è idrolizzato per formare un acido carbossilico e un alcol. O a. CH3 C O [1] −OH OCH2CH3 [2] H3O+ COOCH(CH3)2 b. O C CH3 OH OCH3 [2] H3O+ OH + HOCH3 COOH [1] −OH + HOCH(CH3)2 [2] H3O+ OCH3 O [1] −OH c. + HOCH2CH3 OCH3 20.34 O a. CH3CH2 –OH/H O 2 C 18 OCH3 18 O O CH3CH2 + C H18 b. OCH3 O– CH3CH2 C 18 O – OH/H2O OCH3 CH3CH2 C O CH3CH2 O + HOCH3 20.35 CH2OCO(CH2)15CH3 CH2 OH Na+ −OOC(CH2)15CH3 CH Na+ −OOC(CH2)15CH3 hydrolysis CHOCO(CH2)15CH3 CH2OCO(CH2)7CH=CH(CH2)7CH3 OH Na+ −OOC(CH2)7CH=CH(CH2)7CH3 CH2 OH cis glycerol cis soap 20.36 O O NH H2O/ OH OH O O N H H O O NH NH2 20.37 NaCN a. CH3CH2CH2 Br CH3CH2CH2 CN H2O/–OH c. CN CN COOH H2O/H+ b. COO CN COOH 20.38 a. CH3 O OH NH C C C NH2 O b. 20.39 C CH3 c. NH OH NHCH3 C NCH3 CH3CH2 OH NH2 CH3CH2 C O C 18 O Fondamenti di chimica organica Janice Gorzynski Smith Copyright © 2009 – The McGraw-Hill Companies srl a. CH3CH2 Br [1] NaCN [2] LiAlH4 [3] H2O CH3CH2 b. CH3CH2CH2 CN CH2NH2 O [1] DiBAL–H CH3CH2CH2 C [2] H2O H 20.40 Br NaCN CN H3O+ COOH SOCl2 A COCl [1] (CH3)2CuLi C [2] H2O O D B [1] LiAlH4 CH3 [2] H2O CH3 E OH/H+ PCC CH2NH2 CO2CH3 [1] DIBAL–H CHO [1] CH3Li [2] H2O C OH [2] H2O G F H [1] LiAlH4 (CH3CO)2O [2] H2O CH2OTs NaCN CH2OH CH2NHCOCH3 CH2CN TsCl/pyridine I K J L [1] CH3MgBr [2] H2O O M 20.41 O H D C CH3 a. CH3COCl OH c. O pyridine CH3 CH3 b. Br H D 20.42 CH3 NaCN C H D CH3CH2OH H+ COOH CH3 O CN D H d. CH3 C CH3 + Cl C H 6 5 C H NH2 (2 equiv) C CH3 C6H5 C COOCH2CH3 CH3 C H + H C6H5 NH NH3 + C Cl– CH3 O Fondamenti di chimica organica Janice Gorzynski Smith Copyright © 2009 – The McGraw-Hill Companies srl O a. Cl Cl Cl CH3NH excess O C c. NHCH3 Cl O O b. O CH3NH2 C Cl O O H2O excess Cl HO C + HCl OH Reaction with H2O vapor in the lungs results in the formation of HCl, increasing the acidity of the lungs. HOCH2CH2OH C C O All H's on these C's are identical. 20.43 O H 2N H N H H COOH O H 2O OCH3 H 2N H O H OH H2N OH + CH3OH O COOH phenylalanine aspartame 20.44 O C a. Cl O C Cl C Cl NH2NH NH2NH2 b. O NHNH2 + Cl + HN N HO C NH2NH H O O O O O O O O HO OH O OH O c. H–OSO3H OH OH OH OH OH HO HO OH O H OH H–OSO3H HO OH2 O O – + HSO4– HSO4 + H2SO4 HSO4– O H2SO4 + O O H O + H2O 20.45 Fondamenti di chimica organica Janice Gorzynski Smith Copyright © 2009 – The McGraw-Hill Companies srl H+ O CH3 C OH OH C CH3 OH OH CH3 OH CH3 C OH H OH C OH2 CH3 18 O H O H 18 CH3 18 18 + H2O OH H+ C OH 18 O H O H A + H3O+ + H2O + H2O Two possibilities for A: CH3 OH O H C C CH3 18 O H C O H2O 18 CH3 OH C 18 + H3O+ OH A CH3 OH OH C C CH3 18 O H OH 18 O H CH3 H2O C 18 + H3O+ O A 20.46 O C OH H OSO3H C OCH3 OCH3 OH C OCH3 C OCH3 OCH2CH2CH2CH3 OCH2CH2CH2CH3 H OSO3H H CH3CH2CH2CH2OH + HSO4– OH HSO4 HSO4 O C O H OCH2CH2CH2CH3 + H2SO4 C C OCH2CH2CH2CH3 OCH2CH2CH2CH3 + CH3OH 20.47 OH H OCH3 + HSO4– Fondamenti di chimica organica Janice Gorzynski Smith Copyright © 2009 – The McGraw-Hill Companies srl O R O C – O R' + OH H2O SN2 This bond is not broken. H CH3 O CH3 C O R CH2CH3 X C mechanism CH3 C + R'OH O H CH3 O accepted C R HO O H CH3 O C CH2CH3 (R)-2-butanol CH3 C O X C CH2CH3 According to the accepted mechanism, the stereochemistry around the stereogenic center is retained in the product. This bond is cleaved. H CH3 O CH3 C C O – OH CH3 H2O SN2 alternative CH2CH3 X CH3 H O C O CH3CH2 C OH (S)-2-butanol This SN2 mechanism would form the product of inversion leading to (S)-2-butanol. Since (R)-2-butanol is the only product formed, the SN2 mechanism does not occur during ester hydrolysis. 20.48 Reaction in base: O C CH3 O C O NH2 C O NH H A O CH3 or O C Reaction in acid: O N H B C H A CH3 OH N H A Intermediated X can be re-converted to starting material B or go on to form A by a stepwise process. O H A C CH3 NH C O C CH3 CH3 poorer leaving group O O C OH NH CH3 O O NH3 20.49 C NH CH3 Once this product forms it cannot revert to B since RNH3+ no longer has a lone pair so it is not a nucleophile. N H CH3 B X O C O This product is favored since the negatively charged O atom is a weaker base and therefore better leaving group than the negatively charged N atom. CH3 N H O OH C N H Once this intermediate forms, there are two possibilities. B OH O O N H OH O CH3 NH2 CH3 OH A O CH3 C CH3 O H NH2 O NH2 A C OH H A C O CH3 H A This NH2 is more basic than the OH group in B, so it is protonated in acid. A Fondamenti di chimica organica Janice Gorzynski Smith Copyright © 2009 – The McGraw-Hill Companies srl O OCH2CH3 NH2 C OCH2CH3 O NH2 OH + CH3CH2O C O OCH2CH3 O H + HB+ B X diethyl carbonate (any base) O O HN O OCH2CH3 NH2 C OCH2CH3 O + CH3CH2O– HN B OCH2CH3 H O H N O O NH2 C OCH2CH3 OCH2CH3 O O + HB+ Y + CH3CH2O– 20.50 sp3 C sp2 C O RCH2 Cl less electrophilic C more crowded C since it is surrounded by four atoms R C O Cl R more electrophilic C due to electron withdrawing O more reactive C Cl This resonance structure illustrates how the electronegative O atom withdraws more electron density from C. The sp2 hybridized C of RCOCl is much less crowded, and this makes nucleophilic attack easier as well. 20.51 L'esterificazione di Fischer è il trattamento di un acido carbossilico con un alcol in presenza di un catalizzatore acido per formare un estere. a. (CH3)3CCO2CH2CH3 (CH3)3CCOOH + HOCH2CH3 b. O OH O 20.52 OH O c. O O HO d. O OH O O HO HO O Fondamenti di chimica organica Janice Gorzynski Smith Copyright © 2009 – The McGraw-Hill Companies srl NH3 Br excess a. NH2 H3O+ NaCN Br H N OH NH2 CN O DCC O NaOH b. Br OH H2SO4 OH OH O O [from (a)] O [1] c. CN [from (a)] MgBr Mg Br [2] H2O MgBr O O H2SO4 CrO3 d. OH [from (b)] OH H2SO4/H2O O 20.53 – a. Br CN H2 CN O/H+ COOH CN b. [from (a)] SOCl2 c. COOH COCl [from (b)] d. [from (b)] e. COOH HOCH2CH3 CO2CH2CH3 H2SO4 [1] CH3MgBr [from (a)] CN C [2] H2O CH3 O [1] DIBAL–H f. CN [from (a)] g. CN CH2NH2 [2] H2O h. CH3COOH CH2NH2 [from (g)] 20.54 CHO [2] H2O [1] LiAlH4 [from (a)] O OH DCC CH2NHCOCH3 Fondamenti di chimica organica Janice Gorzynski Smith Copyright © 2009 – The McGraw-Hill Companies srl CN CH3 + NaCN a. CH3Cl H3O+ CH3 COOH [1] CO2 CH3 MgCl Cl + Mg CH3 Br CH3 COOH This method can't be used because an SN2 reaction can't be done on an sp2 hybridized C. + NaCN b. [2] H3O+ sp2 MgBr Br + Mg COOH [1] CO2 [2] H3O+ This method can't be used because an SN2 reaction can't be done on a 3° C. c. (CH3)3CCl + NaCN [1] CO2 (CH3)3C MgCl (CH3)3C Cl + Mg d. HOCH2CH2CH2CH2Br HOCH2CH2CH2CH2 + NaCN H3O+ CN HOCH2CH2CH2CH2 COOH This method can't be used because you can't make a Grignard reagent with an acidic OH group. Br + Mg HOCH2CH2CH2CH2 (CH3)3C COOH [2] H3O+ 20.55 O CH3Cl CH3Cl AlCl3 AlCl3 CH3 COOH KMnO4 CH3 HOCH2CH2CH2CH3 C OCH CH CH CH 2 2 2 3 H2SO4 C OCH2CH2CH2CH3 COOH O dibutyl phthalate 20.56 CH3CH2Cl a. AlCl3 OH COH KMnO4 salicylamide NO2 H2/Pd/C HO HO (+ ortho isomer) H N c. C CH3 O HO acetaminophen [from (b)] 20.57 H N NaH O OH OH NH2 H N CH3COCl H2SO4 OH CNH2 NH3 Cl OH (+ para isomer) b. SOCl2 2 equiv OH HNO3 O O O C CH3 acetaminophen H N CH3CH2Br O CH3CH2O CH3 O HO (More nucleophilic NH2 reacts first.) C C CH3 O p-acetophenetidin Fondamenti di chimica organica Janice Gorzynski Smith Copyright © 2009 – The McGraw-Hill Companies srl CH3Cl a. HCl CH3OH Br2 CH3 AlCl3 CH2Br hν NaCN H2O H+ CH2CN CH2COOH HOCH2CH3 H2SO4 CH3Cl ZnCl2 CH2COOCH2CH3 ethyl phenylacetate CH3 CH3Cl b. NH2 NO2 [from (a)] CH3 HNO3 H2SO4 AlCl3 NH2 CH3 H2 NH2 COOH COOCH3 HOCH3 KMnO4 H2SO4 Pd methyl anthranilate (+ para isomer) [from (a)] O CH3 CH3Cl AlCl3 c. CH3CH2OH COOH KMnO4 OH CH3COOH H2SO4 [1] LiAlH4 [2] H2O O benzyl acetate CrO3 H2SO4/H2O CH3COOH 20.58 [1] LiAlH4 Cl Cl –CN CN NC excess NH2 H2N [2] H2O O H2O OH HO H2SO4 O 20.59 O a. CH3 C OH H2SO4 b. CH313CH2OH CrO3 H2SO4/H2O c. CH3CH2Br O CH313CH2OH H218O d. CH313CH2OH 13 OCH2CH3 O O 13 CH3 C CH3CH2OH OH 18OH + base (H18O–) PBr3 C CH3 CH3CH2 CH313CH2Br – H2SO4 CH3 OCH2CH3 O CH3COCl CH3 C 18 OCH2CH3 18 18 OH 13C CH313CH218OH CrO3 18O O 13 C 18 H2SO4/H218O CH3 OH CH3CH2OH H2SO4 13 C CH3 + OCH2CH3 H218O
Scaricare